Description
1. Product Overview
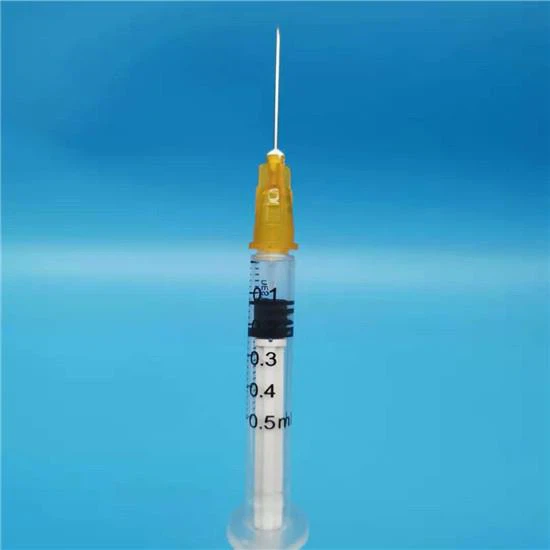
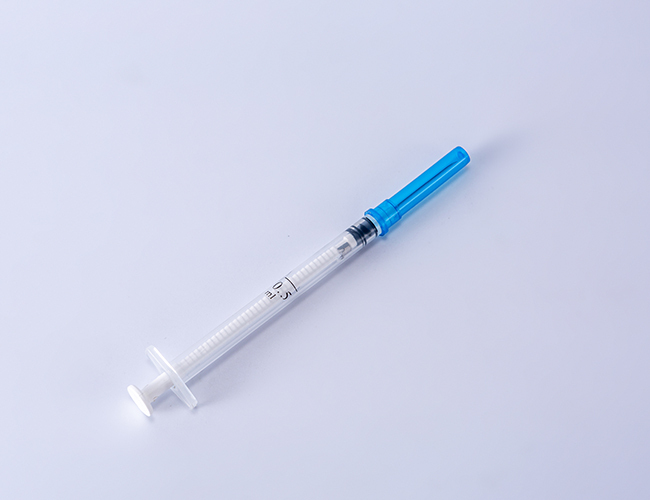
The Auto Disable Syringe 0.5ml is a fixed-dose, single-use injection device engineered specifically for mass immunization campaigns and routine vaccination programs. Its primary industrial application lies in public health supply chains, where preventing syringe reuse is critical to reducing bloodborne pathogen transmission. The key value proposition is the syringe’s integrated auto-disable (AD) mechanism, which locks the plunger after a single complete injection, rendering the device non-reusable. Strategically, this product is indispensable for global health organizations (WHO, UNICEF, Gavi) and national ministries of health seeking to comply with stringent safety standards, reduce cross-contamination risks, and achieve herd immunity targets without iatrogenic disease spread.
2. Key Specifications & Technical Characteristics
-
Material Components: Medical-grade polypropylene (barrel and plunger), thermoplastic elastomer (gasket/stopper), and stainless steel cannula (27G x ½” or 25G x 5/8” options)
-
Safety Mechanism: Passive auto-disable lock-out ring – engages irreversibly after full plunger depression at 0.5ml dose completion
-
Physical Characteristics: Translucent barrel with bold black graduation markings; low dead space design (<0.02ml residual); luer slip or fixed needle configuration
-
Sterility: Ethylene oxide (EtO) sterilized; sterile fluid path guaranteed
-
Packaging Options: Blister-packed individual units (100 pieces per inner box; 2,000 pieces per export carton); peel-open pouch optional
-
Shelf Life: 5 years from date of manufacture when stored in original, unopened packaging at 5°C–40°C (<80% relative humidity)
3. Core Industrial Applications
-
Primary Industries: Public health agencies, NGOs (e.g., IFRC, MSF), vaccine manufacturers (fill-and-finish partners), and government medical logistics depots
-
Operational Use Cases: High-volume vaccination drives (measles, polio, yellow fever, COVID-19 boosters), routine EPI (Expanded Programme on Immunization) schedules, and outbreak response zones with limited waste management infrastructure
-
Performance vs. Alternatives: Unlike standard syringes that require sharps containers and manual oversight to prevent reuse, the AD mechanism delivers passive compliance – no user training required to disable the syringe. This reduces reuse risk from ~30% (observed in low-resource settings) to near zero. Cost-per-dose is 15–20% lower than retractable needle syringes, while maintaining comparable injection quality (low break-loose and gliding forces).
-
Efficiency Advantage: 0.5ml fixed-dose eliminates dosing errors; rapid plunger return break reduces cycle time for vaccinators by ~1.5 seconds per patient compared to adjustable syringes.
4. Competitive Advantages
-
Quality Consistency: ISO 7886-4:2018 certified; lot-release testing meets WHO PQS (Performance Quality and Safety) code E008; 100% leak and mechanical lock integrity inspection prior to packaging
-
Supply Reliability: Dual manufacturing sites (ASEAN + MEA) with 6-month rolling safety stock; audited supply chain with full traceability from resin lot to delivery note
-
Logistics Capability: Compact nesting design – 35% higher unit density per carton than leading competitors, reducing containerized freight cost per 10,000 syringes by ~18%
-
Price Competitiveness: Tiered pricing starting at 0.028–0.035 per unit (FOB) for annual volumes >5 million units; cost includes AD validation certificate per batch
-
Sustainability: 100% virgin PP with recyclability where waste-to-energy infrastructure exists; reduced packaging plastic by 22% (2024 redesign)
-
Technical Support: 24/7 multilingual quality documentation access (COAs, stability studies, regulatory dossiers); on-site supplier audit available for multilateral tenders
5. Commercial & Supply Information
-
Minimum Order Quantity (MOQ): BULK – 20MT (approx. 1,000,000 units) per single SKU
-
Loading Capacity per 20FT Container: 22MT net (1,100,000 units) – 1,100 export cartons (2,000 units/carton)
-
Loading Capacity per 40FT Container: 26MT net (1,300,000 units) – 1,300 export cartons
-
Lead Time: 45 days ARO for MOQ; expedited (25 days) available for 10% premium on existing frame agreements
-
Incoterms preferred: CIF (any major port) or EXW (manufacturing hub)


Reviews
There are no reviews yet.