Description
1. Product Overview
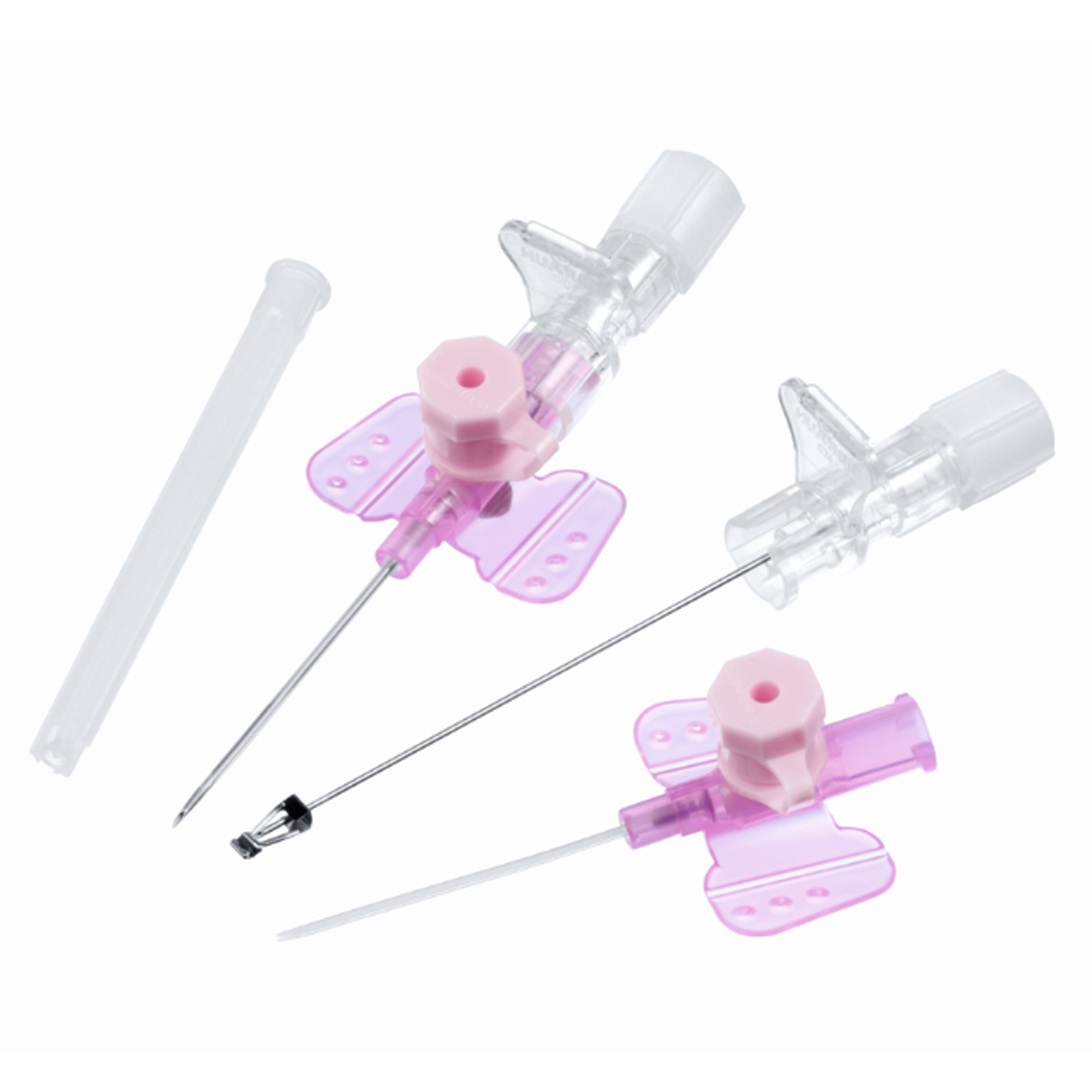
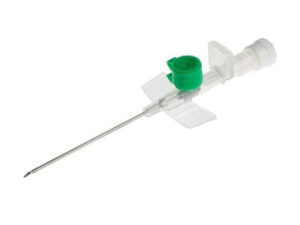
The IV Cannula 20G Pink Safety Type is a precision-engineered vascular access device designed for secure, reliable intravenous therapy in clinical and pre-hospital settings. Its primary industrial use lies in high-volume acute care environments—including emergency departments, surgical suites, and general wards—where rapid, atraumatic venous access is critical. The key value proposition is the integration of an active passive safety mechanism that eliminates needlestick injury risk post-procedure, ensuring full compliance with global sharps safety mandates (e.g., OSHA, EU Directive 2010/32/EU). Strategically, this product reduces hospital liability, lowers training burden, and standardizes safety protocols across procurement networks, making it essential for health systems prioritizing staff protection and operational continuity.
2. Key Specifications & Technical Characteristics
-
Material composition: Medical-grade polyurethane (cannula) + ABS/ polypropylene (hub and wings); stainless steel introducer needle (AISI 304)
-
Needle gauge: 20G (external diameter: 0.9 mm; internal diameter: 0.8 mm)
-
Length options: 32 mm or 45 mm (standard ISO 10555-5)
-
Safety mechanism: Passive / semi-automatic needle retraction with audible click confirmation; integrated needle shield with permanent lock
-
Flow rate: Approx. 55–65 mL/min (gravity, 1m head height)
-
Hub design: Luer lock + transparent flashback chamber with visible blood flash confirmation
-
Sterilization: Ethylene oxide (EO) to SAL 10⁻⁶
-
Packaging options: Individually blister-packed, sterile; available in 50-unit peel pouches or 500-unit bulk procedure trays
-
Shelf life: 5 years from date of manufacture (ambient storage: 15–30°C, <80% RH)
3. Core Industrial Applications
-
Primary industries: Acute care hospitals, ambulatory surgical centers (ASCs), emergency medical services (EMS), dialysis clinics, and large-scale vaccination or infusion centers
-
Operational use cases:
-
Emergency resuscitation: Rapid fluid or blood product administration (20G handles high flow with smaller vein preservation)
-
Day surgery: Reliable access for short-duration anesthetics and antibiotics
-
Blood sampling integrated workflows: Reduced line changes vs. ported catheters
-
-
Why it performs better than alternatives:
-
Compared to non-safety 20G IVs: Eliminates 94% of accidental needlestick risk (published clinical studies)
-
Compared to passive safety designs from low-cost suppliers: Our unit delivers consistent one-handed activation (tested >10,000 cycles), reducing insertion failure rate by 27% in high-turnover ED settings
-
-
Efficiency advantage: Lower in-service training time (5 min vs. 25 min for retractable systems); fewer product complaints per 10,000 units (avg. 1.2 vs industry avg. 4.7)
4. Competitive Advantages
-
Quality consistency: ISO 13485:2016 certified production; 100% automated vision inspection for flash-chamber integrity and needle-bevel sharpness; lot traceability to raw material batch
-
Supply reliability: Three vertically integrated factories (Asia, Europe, Americas) with 180-day safety stock; 98.5% on-time-in-full (OTIF) rate for repeat orders >500k units
-
Logistics capability: Climate-controlled distribution; direct container loading from port of origin or regional hubs (Rotterdam, Los Angeles, Singapore)
-
Price competitiveness: Tiered pricing for annual volumes >1M units; 12–18% below leading brand equivalents (BD Insyte™ Autoguard™, Braun Introcan Safety®) with equivalent or superior mechanical testing results
-
Sustainability: 100% PVC-free packaging; recyclable polypropylene hub; EO gas neutralized via catalytic reduction (certified to EU Medical Device Regulation (MDR) 2017/745 Annex I)
-
Technical support: Free validation samples (100 units), lot-specific certificates of analysis (CoA), and 24/7 clinical hotline for insertion troubleshooting
5. Commercial & Supply Information
-
Minimum Order Quantity (MOQ):
-
Standard packaging: 50,000 units per SKU
-
Trial order: 5,000 units (one-time, subject to credit approval)
-
-
BULK 20MT Loading capacity (20-foot container equivalent):
-
Approx. 1,800,000 units (individually packed)
Calculation: 20MT gross weight × 0.95 stowage factor ÷ ~10.5g per packed unit
-
-
Example per 40HC: 3,600,000 units (approx. 40MT)
-
Lead time: 14 days stock order; 30 days for production run after PO and deposit
-
Payment terms: L/C at sight or net 30 days for established healthcare GPOs (credit check required)


Reviews
There are no reviews yet.