Description
1. Product Overview
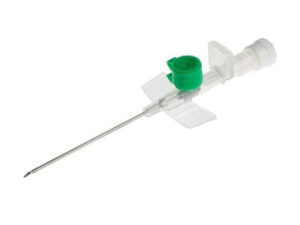
The IV Cannula 16G Grey With Injection Port is a high-flow vascular access device engineered for rapid fluid resuscitation, blood product transfusion, and high-viscosity drug administration in acute care settings. Its primary industrial use lies within hospital systems, emergency medical services (EMS), and surgical centers where large-bore venous access is critical to patient survival. The key value proposition is its optimal balance between internal diameter (1.7 mm) and flow rate (~105 mL/min) — the largest gauge still suitable for peripheral placement in adult trauma patients. Strategically, it is indispensable for operating rooms, ERs, and ICU crash carts, directly impacting mortality reduction in hypovolemic shock scenarios.
2. Key Specifications & Technical Characteristics
-
Material Composition:
-
Cannula: Medical-grade PTFE (polytetrafluoroethylene) – non-thrombogenic, radiopaque line for imaging confirmation
-
Needle: Stainless steel (AISI 304), tri-beveled lancet point for minimal insertion trauma
-
Injection Port: Latex-free silicone septum with positive fluid seal; polycarbonate housing
-
Wings/Flashback Chamber: Clear polypropylene with integrated blood flashback visualization
-
-
Physical Characteristics:
-
Gauge: 16G (Grey international color standard)
-
Internal Diameter: 1.7 mm (+/- 0.05 mm)
-
External Diameter: 2.1 mm
-
Length: 45 mm (standard adult peripheral length)
-
Flow Rate: ≥ 105 mL/min (water at 100 cm head pressure)
-
Bevel Angle: 12–15 degrees
-
Injection Port Lumen: Integral Y-port, luer lock compatible (ISO 594-1)
-
-
Sterility & Grade:
-
Sterilization: Ethylene oxide (EtO) – SAL 10⁻⁶
-
Grade: Class I medical device (CE, FDA 510(k) cleared equivalent)
-
Pyrogen-free, non-cytotoxic, ISO 10993 biocompatibility compliant
-
-
Packaging Options:
-
Blister pack: Individually sealed, peel-open Tyvek/PET
-
Box: 50 units/box (sterile, shelf-ready)
-
Case: 500 units (10 boxes) per case – standardized for hospital logistics
-
-
Shelf Life: 5 years from date of manufacture (unopened, controlled storage: 10–30°C, RH < 60%)
3. Core Industrial Applications
-
Primary Industries:
-
Acute care hospitals (ED, ICU, OR, trauma bays)
-
Pre-hospital EMS (ambulance, helicopter, field hospitals)
-
Combat medical logistics (military battlefield medicine)
-
Dialysis centers (as a temporary access bridge)
-
-
Specific Operational Use Cases:
-
Massive transfusion protocol (MTP) – whole blood or packed RBCs (16G avoids hemolysis vs. smaller gauges)
-
Rapid crystalloid infusion (e.g., 1L lactated Ringer’s in < 5 min)
-
CT contrast injection (high viscosity, 5–8 mL/sec)
-
Vasopressor administration (via injection port without interrupting primary line)
-
-
Performance vs. Alternatives:
-
Vs. 18G (green): 40% higher flow rate (critical in shock).
-
Vs. 14G (orange): Lower failure rate on first attempt (fewer venous blowouts), longer dwell time (7 days vs. 3–4 days), and safer for conscious patients.
-
Vs. non-port cannulas: Injection port eliminates need for extra Y-connector, reducing contamination risk and setup time by ~30 sec per line change.
-
Cost advantage: Reduced catheter replacements (less phlebitis due to PTFE) and fewer staff needlesticks (integrated safety mechanism available on request).
-
4. Competitive Advantages
-
Quality Consistency: Batch-to-batch variability < 0.5% on inner diameter and flashback speed. Each unit undergoes automated leak testing (injection port to 300 kPa) and needle tip integrity inspection (200x magnification visual AI system).
-
Supply Reliability: Dual manufacturing sites (ISO 13485 certified) with 6-month rolling safety stock. Lead time guarantee: ≤ 30 days for container orders. Disaster response surge capacity: +200% within 10 days.
-
Logistics Capability: Pre-cleared customs documentation for 50+ countries (including EU, UAE, Saudi Arabia, Singapore, Brazil). Temperature-controlled shipping available (2–8°C not required, but optional for cold-chain integration).
-
Price Competitiveness: Tiered pricing structure — 18–25% below leading brand (B. Braun/BD) for equivalent specifications. Volume discounts activate at 20MT (approx. 20 million units).
-
Sustainability: PTFE recyclable via pyrolysis partner network. Reduced sterile barrier packaging (20% less plastic vs. standard blister). EtO neutralization protocol meets EU ETS standards.
-
Technical Support: Free downloadable IFU (Instructions for Use) in 12 languages. 24/7 clinical application engineering support. Lot traceability via GS1-128 barcode on each box.
5. Commercial & Supply Information
-
Minimum Order Quantity (MOQ):
BULK 20MT (metric tons)
*Equivalent to approximately 20 million units (based on 1 gram/unit cannula weight + sterile packaging).*
Single lot – same batch number for full traceability. -
Loading Capacity per 20FT Container:
20MT net weight-
Palletized: 20 standard euro-pallets (80 x 120 cm), stacked 1.8m high
-
Configuration: 500,000 units per pallet → 10 million units total per 20FT container
-
Note: Two 20FT containers required for full 20MT MOQ.
-
-
Incoterms Offered: FOB (Shanghai/Hamburg) or CIF (major global port).
-
Payment Terms: LC at sight or 30% TT deposit / 70% against BL copy (for verified buyers with credit check).
For strategic sourcing inquiries and sample lot (10,000 units) requests, initiate contact through marketplace RFQ channel.



Reviews
There are no reviews yet.