Description
1. Product Overview
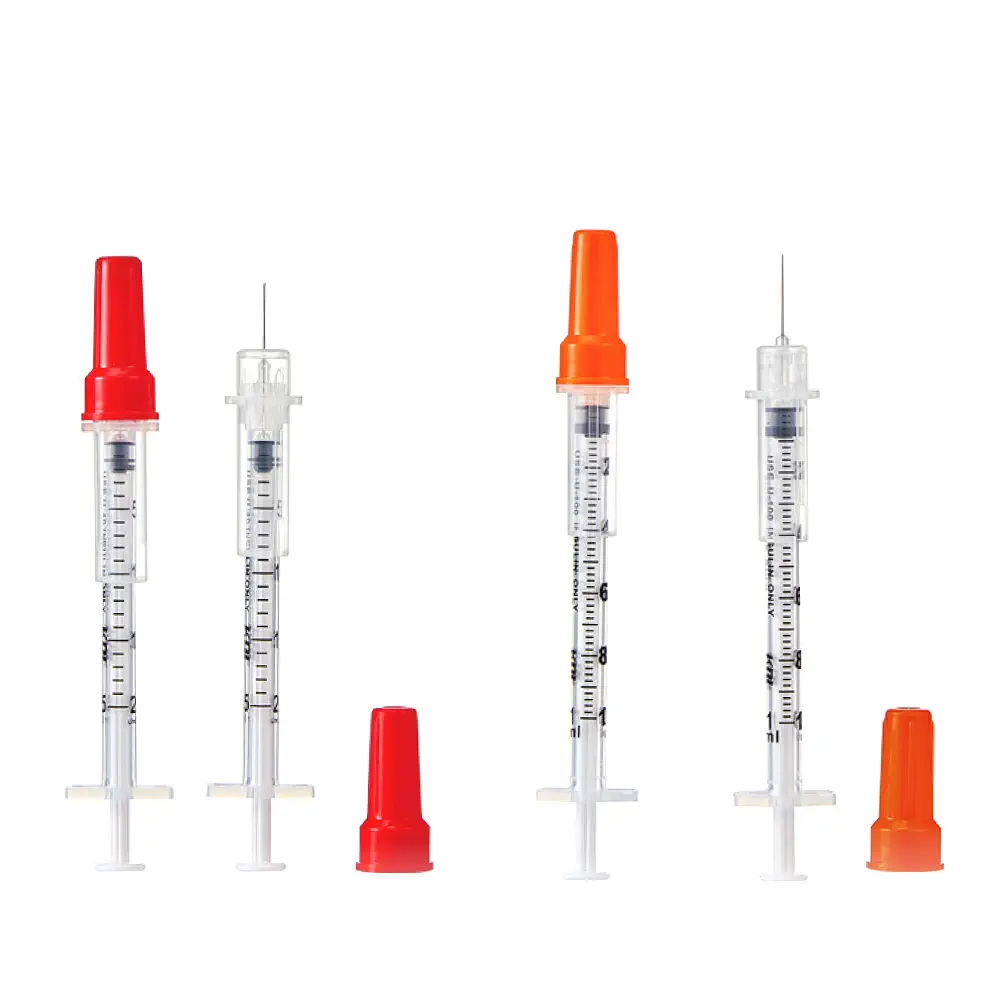
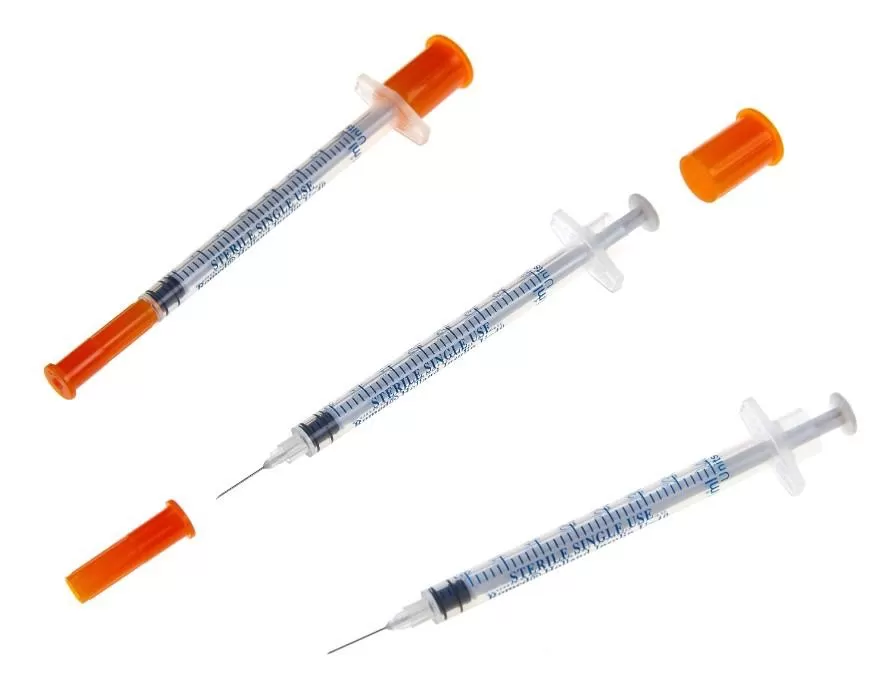
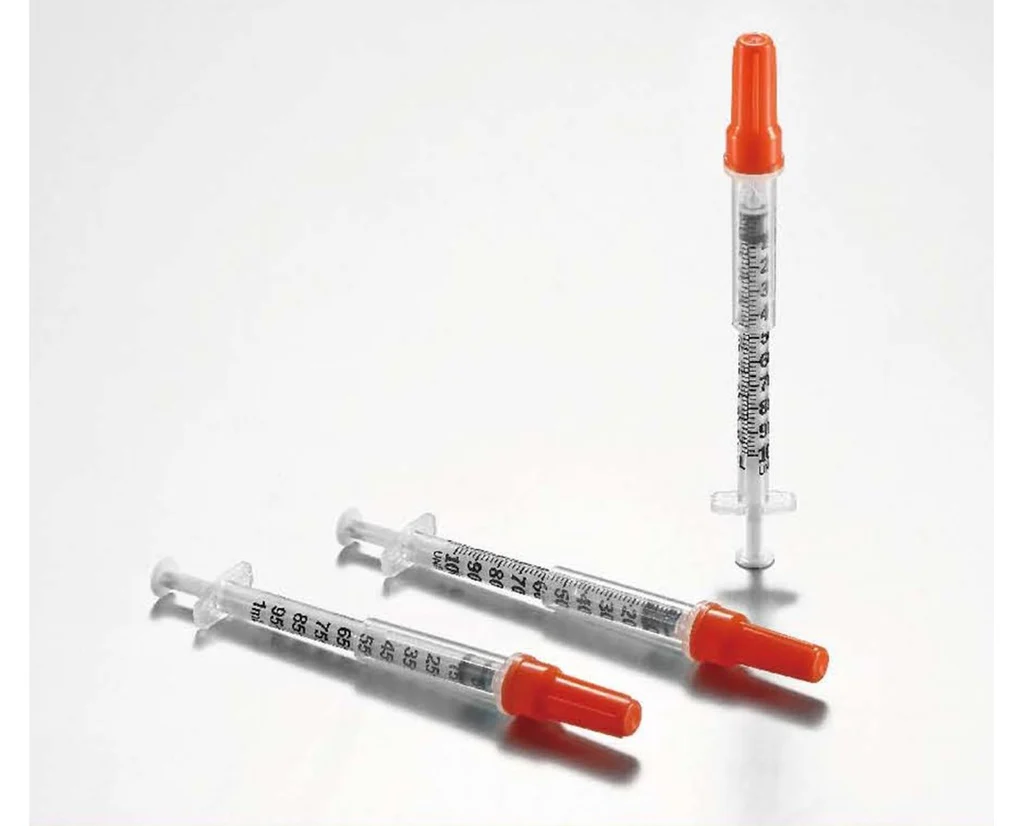
The Insulin Syringe 1ml U-100 Fixed Needle is a precision-engineered medical device designed for the accurate subcutaneous administration of U-100 insulin, primarily indicated for the management of diabetes mellitus in clinical and home-care settings. Its primary industrial use lies within the high-volume manufacturing and distribution ecosystems serving hospitals, diabetes care clinics, retail pharmacies, and institutional healthcare providers. The key value proposition is uncompromised dosing accuracy combined with reduced dead space to minimize medication waste and cross-contamination risk. Strategically, this product is critical in global markets due to the rising prevalence of diabetes and the corresponding demand for affordable, reliable, single-use insulin delivery systems that enhance patient safety while meeting stringent regulatory standards.
2. Key Specifications & Technical Characteristics
-
Material Components: Medical-grade polypropylene (barrel & plunger), stainless steel (29G–31G needle), thermoplastic elastomer (gasket); latex-free, DEHP-free.
-
Graduation Scale: U-100 (100 units per 1ml), with clear, permanent, non-smudge markings in 1-unit or 2-unit increments.
-
Needle Gauge Range: 29G to 31G (ultra-thin wall options available)
-
Needle Length: Fixed 6mm, 8mm, or 12.7mm (1/2 inch) – standard options
-
Dead Space: Low (<8µl) – engineered for fixed-needle configuration
-
Packaging Options: Individually blister-packed; available in 10’s, 100’s (box), or bulk sterile nested trays (1,000 units per inner carton).
-
Shelf Life: 5 years from date of manufacture (when stored in controlled conditions: 5°C–30°C, <80% RH)
-
Sterilization: Ethylene oxide (EtO) to SAL 10⁻⁶; compliant with ISO 11135
3. Core Industrial Applications
-
Primary Industries: Diabetes device manufacturing, hospital central supply chains, long-term care facilities, home healthcare logistics, and government immunization/diabetes control programs.
-
Specific Operational Use Cases:
-
Hospital insulin pen-alternative protocols: Used for single-patient, multiple daily injections (MDI) with fixed needle to reduce needlestick injuries.
-
Bulk distribution for diabetes clinics: Enables rapid, error-reduced insulin draws from multidose vials.
-
Emergency & disaster medical kits: Preferred over removable-needle syringes due to lower risk of needle detachment during transport.
-
-
Performance Advantages vs. Alternatives:
-
vs. Removable needle syringes: Lower dead space reduces insulin waste by ~15–20% – critical for high-cost analog insulins.
-
vs. Insulin pens: More cost-efficient for institutional formularies and adaptable to any brand of U-100 vial.
-
Durability: Fixed needle eliminates loosening or leakage during injection, ensuring consistent dose delivery.
-
4. Competitive Advantages
-
Quality Consistency: ISO 13485:2016 certified production with 100% automated vision inspection for barrel clarity, needle patency, and scale accuracy. Lot-to-lot coefficient of variation (CV) <2% for fill volume.
-
Supply Reliability: Dual manufacturing sites (ASEAN + LATAM) with redundant supply chain; typical lead time 30 days for orders up to 40 containers; consignment inventory available for tier-1 distributors.
-
Logistics Capability: Pre-validated for ambient sea freight and temperature-controlled air freight. Optimized carton dimensions (60x40x30cm) fit standard pallet footprints.
-
Price Competitiveness: Tiered pricing beats EU and US incumbents by 18–25% at comparable quality (CE-MDR, UKCA, and WHO PQS prequalified options available).
-
Sustainability: Transitioning to FSC-certified paper packaging and hospital-recyclable polypropylene; valid EoI for EPR compliance in EU 2026 roadmap.
-
Technical Support: Full regulatory dossiers (STED format), stability data, and biocompatibility reports (ISO 10993) provided under NDA.
5. Commercial & Supply Information
-
Minimum Order Quantity (MOQ): Bulk – 20 metric tons (MT) per product line/item.
-
Loading Capacity (MT per container):
-
20 ft standard container: Approx. 12 MT (dependent on blister vs. bulk packing density)
-
40 ft high-cube container: Approx. 24 MT
-
Note: For 20 MT MOQ, shipment default is one 40 ft HC container.
-
-
Lead Time for MOQ: 45 days after confirmed L/C or deposit.
-
Incoterms supported: FOB (Ningbo / Rotterdam), CIF, EXW.

Reviews
There are no reviews yet.