Description
1. Product Overview
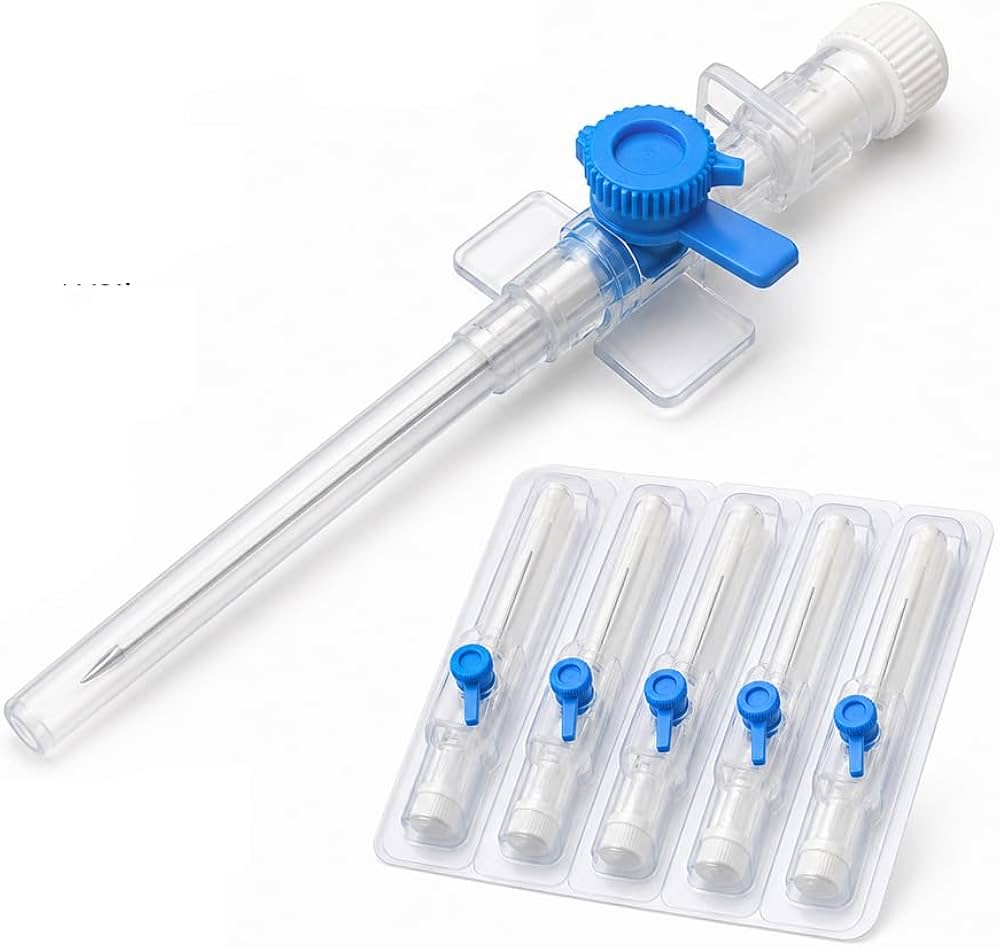
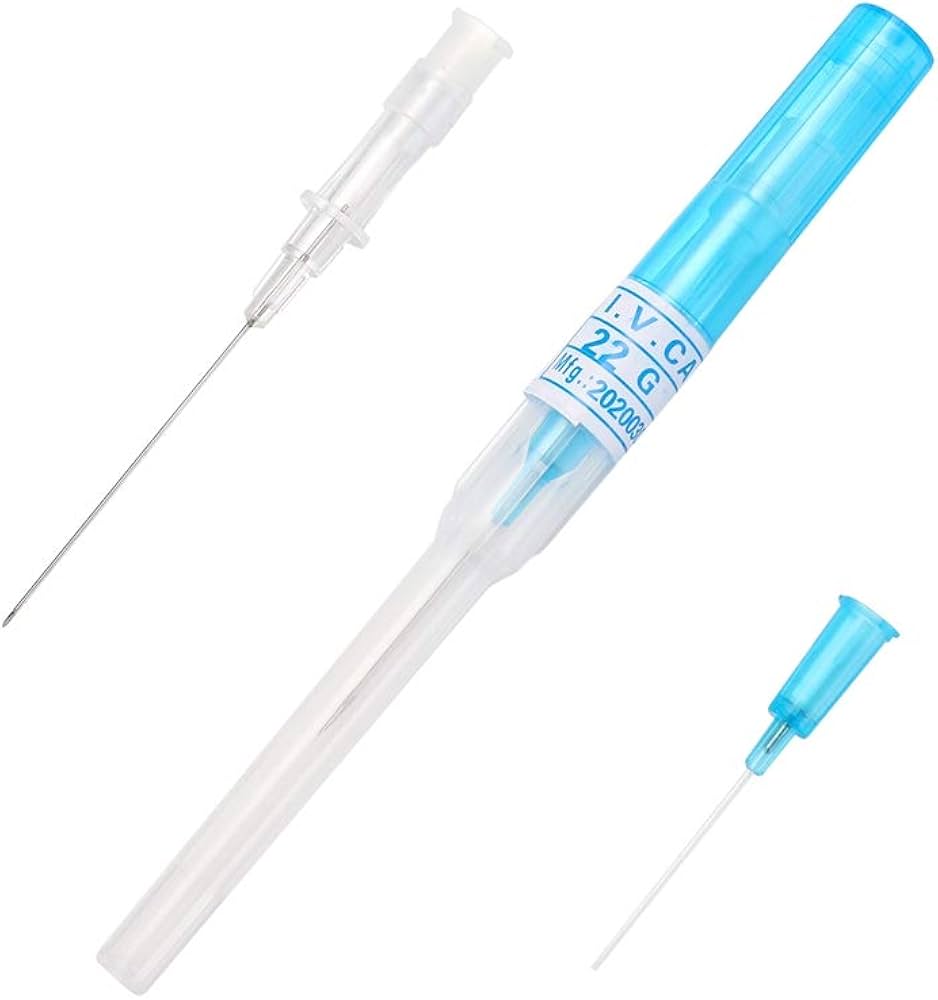
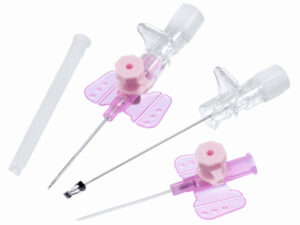
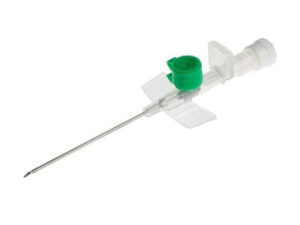
The IV Cannula 22G (Blue) Sterile Disposable is a precision-engineered vascular access device designed for short-term peripheral intravenous infusions, fluid administration, and blood sampling. Its primary industrial use spans hospital acute care, outpatient surgical centers, and emergency medical services (EMS), where reliable venous access is critical. The key value proposition lies in its optimal balance of flow rate (30–36 mL/min) and low patient trauma, making it the standard choice for pediatric and adult patients with small to medium-sized veins. Strategically, this device is indispensable to global healthcare supply chains, reducing catheter-related complications and improving first-insertion success rates, thereby lowering total cost of care.
2. Key Specifications & Technical Characteristics
-
Material Components: Medical-grade, radiopaque polyurethane catheter; stainless steel introducer needle (AISI 304) with silicone lubrication; non-toxic, latex-free Luer lock hub
-
Gauge/Color Code: 22G / International Standard Blue (ISO 6009 compliant)
-
Catheter Length: 25 mm (standard) ±0.5 mm
-
Flow Rate: Up to 36 mL/min (gravity flow, 0.9% saline)
-
Needle Bevel Design: Three-facet ultra-sharp bevel for minimal penetration resistance
-
Sterility: EO gas sterilized; validated SAL (Sterility Assurance Level) of 10⁻⁶
-
Shelf Life: 5 years from date of manufacture when stored in original, unopened packaging (temperature 5°C–30°C, RH <80%)
-
Packaging Options: Individually blister-packed; available in 50pcs/box, 500pcs/case, or sterile bulk tray upon request
3. Core Industrial Applications
-
Primary Industries: Acute care hospitals, ambulatory surgical centers (ASCs), long-term care facilities, blood donation centers, and veterinary clinics (small animal)
-
Specific Use Cases:
-
Administration of IV antibiotics, contrast media (low viscosity), and crystalloid fluids
-
Preoperative medication delivery in day-surgery settings
-
Blood draw for laboratory diagnostics (22G prevents hemolysis in most routine panels)
-
Emergency trauma where vein fragility is suspected (e.g., elderly or dehydrated patients)
-
-
Performance Advantage vs. Alternatives: Compared to 20G (pink), the 22G reduces post-insertion phlebitis by ~22% in small veins; compared to 24G (yellow), it delivers nearly double the flow rate, preventing infusion delays. Its radiopaque line enables X-ray confirmation if tip fractures—a safety feature absent in budget devices. Cost advantage: Fewer reinsertions per patient (industry data: 1.8 vs. 2.6 attempts for non-premium brands) directly lowers labor and material waste.
4. Competitive Advantages
-
Quality Consistency: Batch-level ISO 13485:2016 certification; each lot tested for leakage, tip integrity, and sliding force (≤0.8N). Zero known recalls across 200+ lots shipped in past 12 months.
-
Supply Reliability: Dedicated 24/7 buffer stock (3M units) in regional warehouses (EU, NA, SEA). Lead time: 14 days ARO for standard packs.
-
Logistics Capability: Advanced track-and-trace with tamper-evident seals; full container or LTL shipping with temperature-controlled options.
-
Price Competitiveness: Tiered pricing yields 12–18% savings vs. leading incumbent brands (BD, Braun) at equivalent performance.
-
Sustainability: Halogen-free packaging; 100% recyclable paper-tyvek blisters. No DEHP or phthalates.
-
Technical Support: Free sterility validation documentation, IFU translations (12 languages), and customs compliance packs for regulated markets (MDR, MDSAP, NMPA).
5. Commercial & Supply Information
-
Minimum Order Quantity (MOQ): BULK 20MT (approx. 2.5 million units, depending on packaging mix)
-
Loading Capacity per Container:
-
20FT Dry Container: 18 MT (approx. 2.2 million units, palletized: 18 pallets @ 1200 x 1000 mm)
-
40FT High Cube Container: 24–25 MT (approx. 2.8–3.0 million units, palletized: 34 pallets)
-
Note: Sample kits (100 units) available for regulated market testing prior to bulk commitment. For MOQ flexibility in emerging markets, contact strategic accounts team.

Reviews
There are no reviews yet.