Description
1. Product Overview
The Enteral Feeding Tube 12Fr is a precision-manufactured medical device designed for nasogastric or nasoenteric access, enabling safe and reliable delivery of nutrition, hydration, and medication to patients with compromised oral intake. Its primary industrial use spans acute care hospitals, long-term care facilities, and home healthcare providers requiring high-flow-capacity enteral access in adult and pediatric populations. The key value proposition lies in its optimized 12 French diameter—balancing patient comfort with sufficient lumen size for uninterrupted formula delivery. As global enteral nutrition markets grow at 5–6% annually, standardizing on a clinically validated, MR-conditional tube reduces aspiration risks and unplanned replacement costs, making it a strategic inventory item for integrated delivery networks.
2. Key Specifications & Technical Characteristics
-
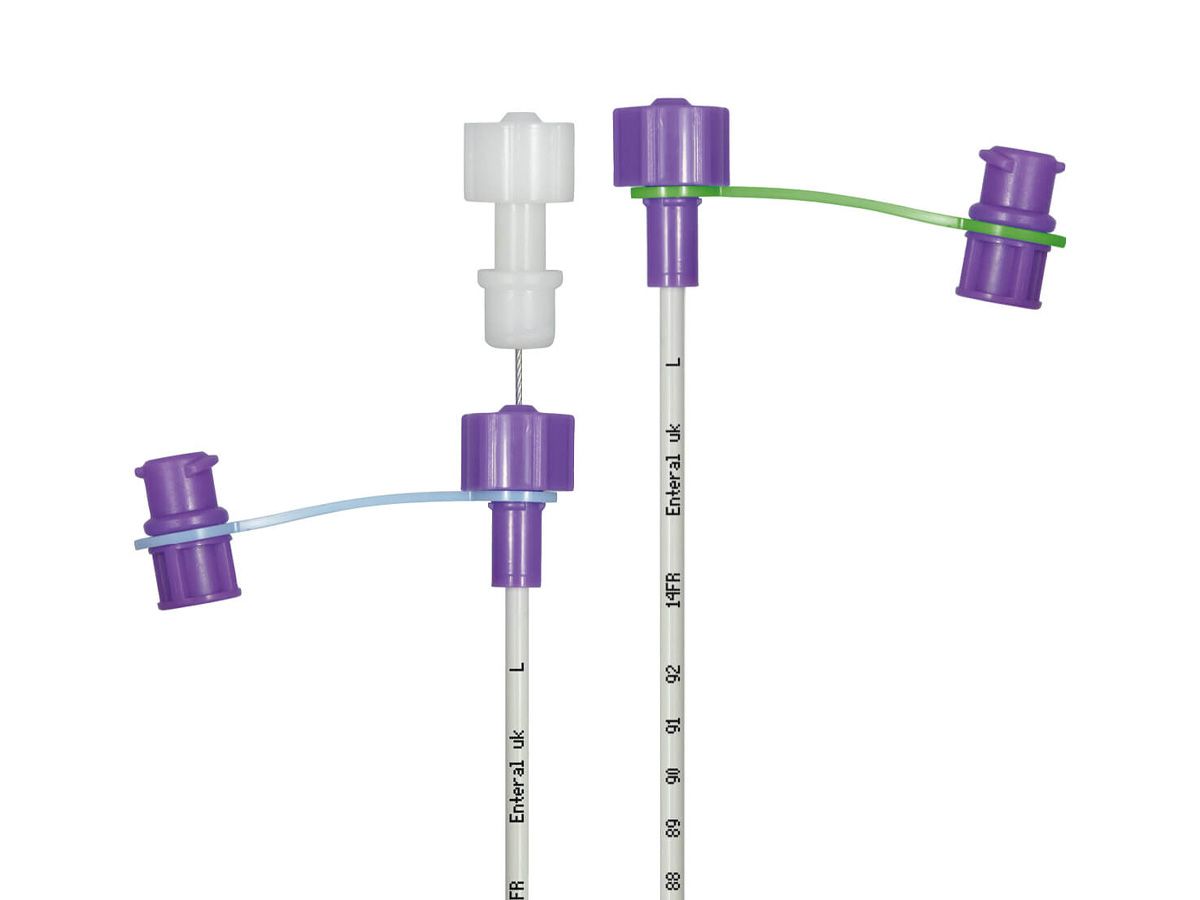
Material Components: Medical-grade polyurethane (PU) – silicone-free; radiopaque barium sulfate stripe integrated; DEHP-free, latex-free.
-
Physical Characteristics:
-
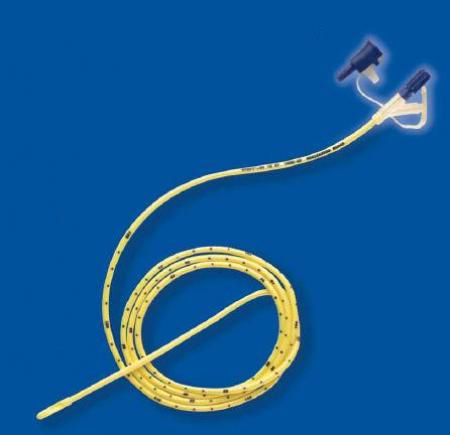
Form: Single-use, sterile, straight tube with rounded closed tip and lateral eyes
-
Color: Transparent with yellow radiopaque indicator line
-
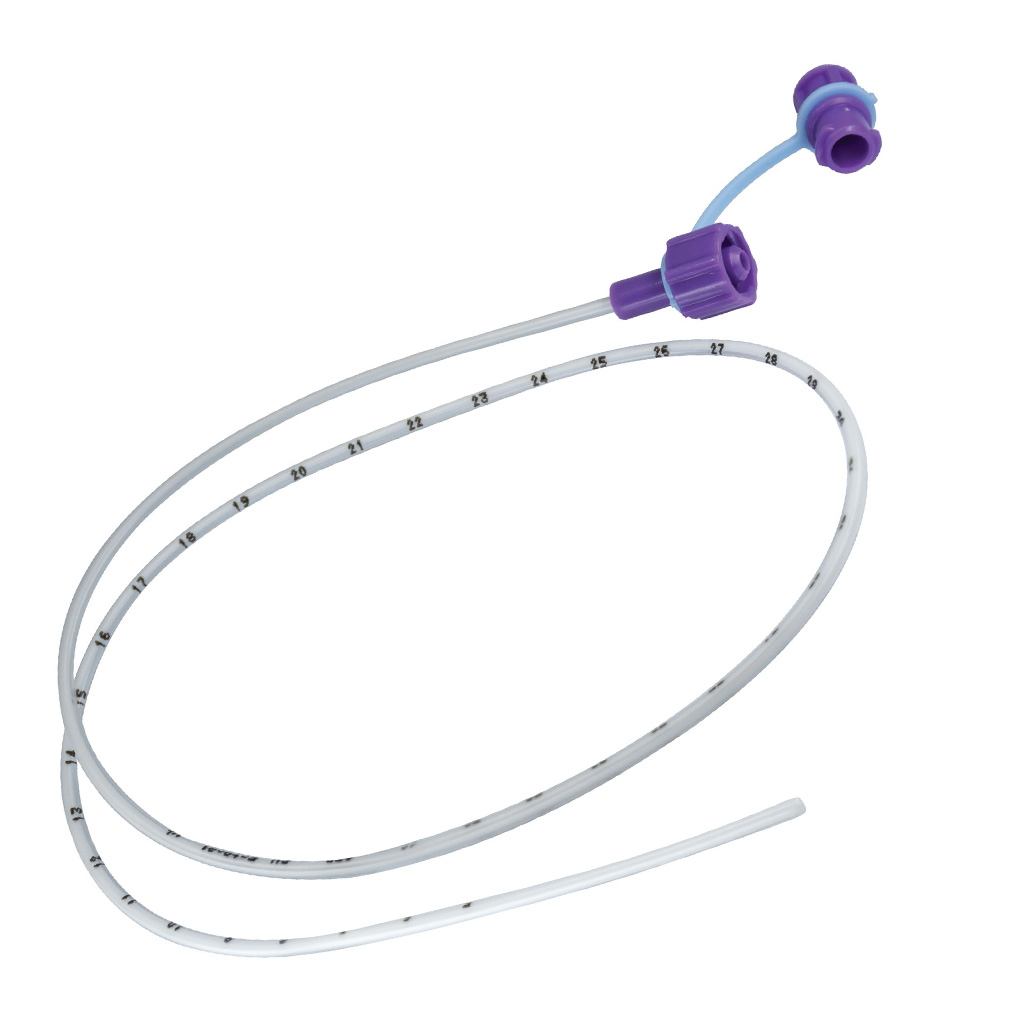
Length: 110 cm (standard adult) ±2 cm
-
Outer diameter: 4.0 mm; Inner diameter: 2.4 mm
-
Stylet: Stainless steel, fully encapsulated, removable
-
-
Packaging Options: Individually sterile-sealed peel pouch; 50 units per master carton; 4 master cartons per case (200 units)
-
Shelf Life: 5 years from date of manufacture (controlled storage: 5–30°C, <60% RH)
-
Certifications: ISO 10993 biocompatibility, CE MDR Class IIa, FDA 510(k) cleared, ISO 13485 manufacturing
3. Core Industrial Applications
-
Primary Industries: Hospital group purchasing organizations (GPOs), skilled nursing facilities (SNFs), home infusion pharmacies, emergency medical services (EMS).
-
Operational Use Cases:
-
Post-stroke dysphagia management – 12Fr enables gravity or pump-assisted feeding without clogging standard 2.0–2.4 mm inner diameter formulas.
-
Post-surgical gastric decompression – Integrated stylet allows rigid placement in sedated patients; MRI conditional up to 1.5 Tesla.
-
Pediatric-to-adult transition units – 12Fr serves as a universal size for patients >10 kg, reducing SKU complexity by 40% compared to stocking 8Fr, 10Fr, and 14Fr separately.
-
-
Performance advantage: Polyurethane material maintains lumen integrity for 30+ days (vs. PVC 7–10 days), reducing replacement frequency by 67%, lowering total cost of ownership per patient episode.
4. Competitive Advantages
-
Quality consistency: Lot-to-lot coefficient of variation <2% on inner diameter and tensile strength; every batch validated via three-point flow resistance testing (water, peptide-based formula, fiber-enriched formula).
-
Supply reliability: Dual-continent manufacturing (Asia + Europe) with 95% on-time-in-full (OTIF) delivery to 40+ countries; 6-month rolling forecast buffer stock held in regional hubs.
-
Logistics capability: Hazard-free medical device classification (non-DG) enables air, sea, and rail shipment with real-time tracking via API integration.
-
Price competitiveness: Volume-based tiering (5,000–50,000 units/month) aligns with GPO contract benchmarks, typically 12–15% below incumbent J&G or Medline equivalents for equivalent specifications.
-
Sustainability: Recyclable polyurethane and paper-based sterile barrier packaging; 30% PCR content in master cartons.
-
Technical support: Full eIFU library, ISO 14971 risk management file, and 24-hour clinical application engineering support for enteral pump integration.
5. Commercial & Supply Information
-
Minimum Order Quantity (MOQ): 10,000 units (mixed lengths accepted – 90 cm, 110 cm, 130 cm)
-
Bulk 20MT Loading Capacity: 1 × 20FT container = 320 master cartons = 64,000 units (160 cartons per pallet × 2 pallets per container layer × 5 layers)
-
Lead time: 20 days ex-works for orders ≤ 64k units; 35 days for ≥ 250k units
-
Incoterms supported: EXW, FOB, CIF (major global ports: Rotterdam, Shanghai, Houston, Dubai)
-
Payment terms: Letter of credit (L/C at sight) for initial order; net 30 days after 3 consecutive compliant shipments (credit approved).




Reviews
There are no reviews yet.