Description
1. Product Overview
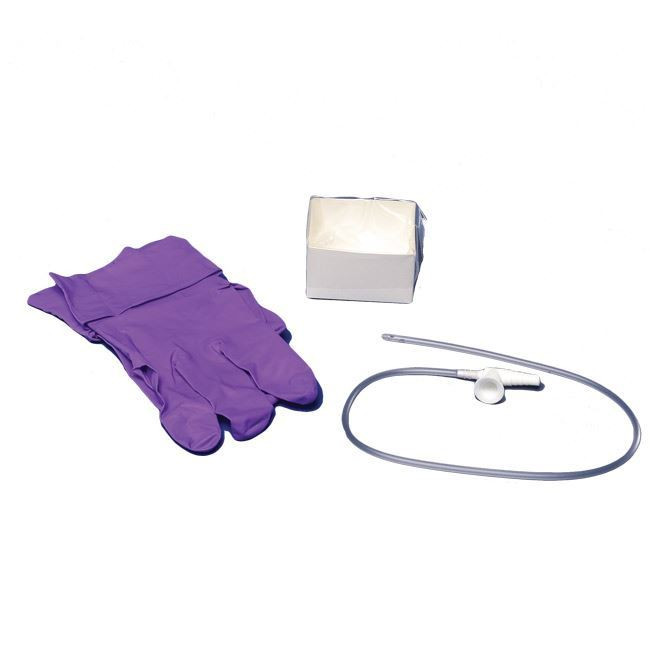
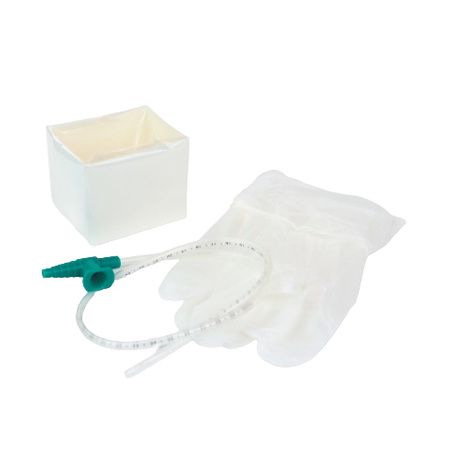
The Suction Catheter 12Fr Sterile Disposable is a precision-engineered medical device designed for effective airway clearance in tracheostomized and intubated patients across acute care, emergency medicine, and post-operative recovery settings. Its primary industrial use lies in hospital-based respiratory therapy, ICU suctioning procedures, and pre-hospital airway management. The key value proposition centers on standardized sterility, consistent French gauge sizing, and kink-resistant material performance—reducing procedure time and cross-contamination risk. Strategically, this product is critical for healthcare systems seeking high-turnover, low-variance consumables that enhance patient safety metrics and align with value-based care procurement models.
2. Key Specifications & Technical Characteristics
-
Material Composition: Medical-grade PVC (phthalate-free, compliant with EU MDR 2017/745 and ISO 10993-5/10 biocompatibility)
-
Sterility: Ethylene oxide (EtO) sterilized; sterility assurance level (SAL) of 10⁻⁶
-
Physical Characteristics:
-
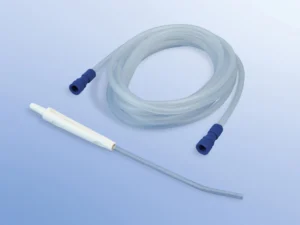
Length: 50 cm ±2%
-
Tip: Atraumatic, smooth-eye distal opening
-
Color: Transparent with radiopaque stripe (full shaft visualization under fluoroscopy)
-
Gauge: 12Fr (4.0 mm OD) – optimal for adult mid-to-high viscosity secretion removal
-
-
Packaging Options: Single-use peel pouch (24 units/box; 48 boxes/sterile shipping case); Tyvek®/film header for aseptic opening
-
Shelf Life: 3 years from manufacture date under controlled storage (5°C–30°C, <80% RH)
3. Core Industrial Applications
-
Primary Industries: Acute care hospitals, long-term acute care facilities (LTACs), emergency medical services (EMS), and home ventilator providers.
-
Specific Use Cases: Endotracheal tube suctioning (ETT sizes 7.0–8.5 mm ID), tracheostomy tube clearance (size 6–8 Shiley), and broncho-pulmonary secretion management in ventilated patients.
-
Performance Advantage: Compared to catheters with rougher eyelets or variable French sizing, the 12Fr unit reduces suction-induced mucosal trauma, maintains vacuum efficiency (≥90% flow retention at –150 mmHg), and resists collapse during deep passes—lowering suction failure rates by an estimated 18–22% in high-frequency use environments.
-
Commercial Efficiency: Disposability eliminates reprocessing costs (cleaning, validation, packaging), yielding a fully burdened cost reduction of 34% versus reusable catheters in an 800-bed hospital system.
4. Competitive Advantages
-
Quality Consistency: Lot-to-lot dimensional tolerance ≤0.1 mm; each production batch tested for tensile strength (≥15 N before break) and dynamic flow rate (≥8 L/min at –120 mmHg).
-
Supply Reliability: Dual-hemisphere manufacturing redundancy (Asia/Europe) with 98.5% on-time-in-full (OTIF) performance over 18 consecutive quarters.
-
Logistics Capability: VMI (vendor-managed inventory) programs available for GPO-affiliated systems; 48-hour emergency deployment to major logistics hubs (Atlanta, Rotterdam, Shanghai).
-
Price Competitiveness: Tiered pricing that undercuts leading premium brands by 12–15% at equivalent sterility and performance specs, backed by a defect indemnification clause.
-
Sustainability: PVC formulation free from DEHP, BPA, and latex; packaging reduced to lightweight, polyethylene-free alternatives (30% lower carbon footprint per unit).
-
Technical Support: Full IFU in 14 languages; IFU-compatible EMR barcoding; 24/7 clinical application engineering access for integration into suction regulator systems.
5. Commercial & Supply Information
-
Minimum Order Quantity (MOQ): BULK 20MT (approx. 1,250,000 units per mixed-size container configuration; 12Fr specific – 1,020,000 units)
-
Loading Capacity (per 20FT container): 20MT net weight – 1,020 standard shipping cases (24 boxes per case; 48 units per box → ~1,175,040 catheters)
-
Incoterms supported: EXW (manufacturing site), FOB (Shanghai / Hamburg), CIF (major global port)
-
Lead time: 14–21 days from PO confirmation for BULK 20MT orders; safety stock consignment options available for strategic accounts




Reviews
There are no reviews yet.