Description
1. Product Overview
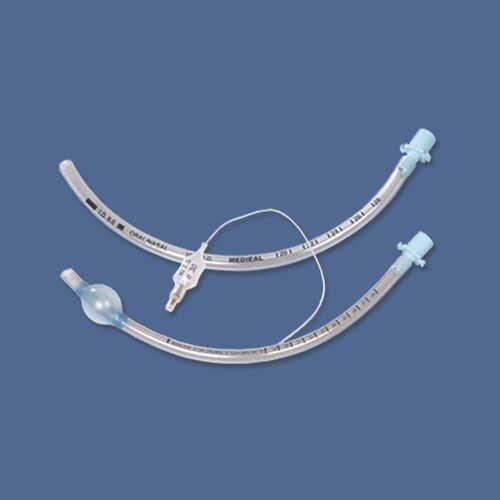
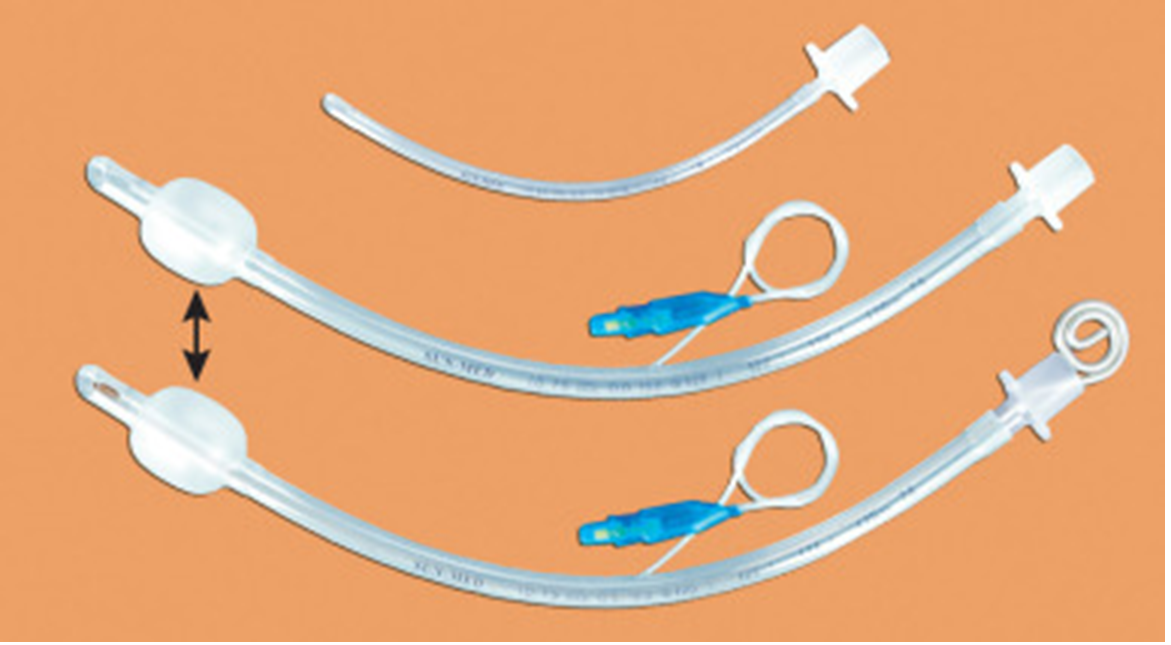
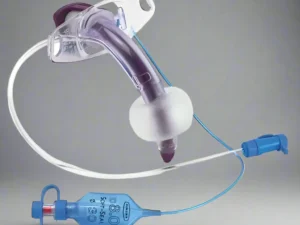
This Endotracheal Tube (ETT) Cuffed 8.0mm with High Volume Low Pressure (HVLP) technology is a Class II medical device engineered for definitive airway management in perioperative and critical care settings. Its primary industrial use lies in mechanical ventilation for adult patients requiring secured airway patency during general anesthesia, respiratory failure, or emergency resuscitation. The key value proposition is the reduction of post-intubation tracheal morbidity: the HVLP cuff design maximizes mucosal surface contact while minimizing lateral wall pressure, thereby lowering the risk of ischemic injury. Strategically, this product is critical for hospital systems and EMS providers seeking to balance patient safety outcomes with cost-efficient, reliable supply chain integration.
2. Key Specifications & Technical Characteristics
-
Material Composition: Medical-grade, non-cytotoxic Polyvinyl Chloride (PVC), DEHP-free (upon requested variant); radiopaque line embedded (barium sulfate).
-
Cuff Type: High Volume, Low Pressure (HVLP) – compliant, thin-walled design.
-
Physical Characteristics:
-
Size: 8.0 mm internal diameter
-
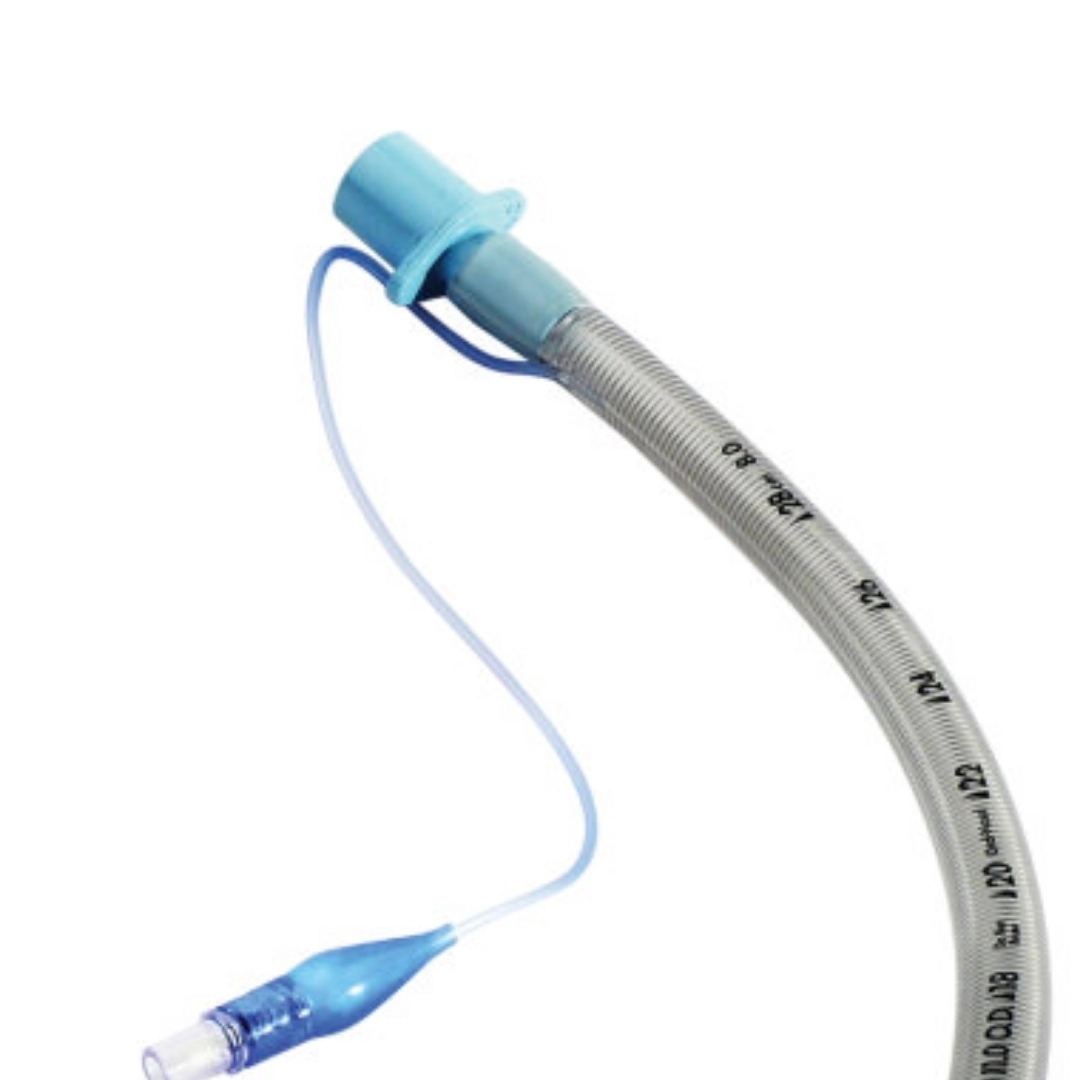
Color-coded 15 mm connector (standardized ISO 5356-1)
-
Smooth, atraumatic bevelled tip with left-facing Murphy eye
-
Pilot balloon with one-way valve (Luer lock compatible)
-
-
Packaging Options: Individually sealed sterile blister pack; 10 units per inner carton; 50 units per master case (hospital bulk pack available upon request).
-
Shelf Life: 5 years from date of manufacture, when stored in controlled environment (10°C–30°C / 50°F–86°F, <70% relative humidity, away from direct sunlight).
3. Core Industrial Applications
-
Primary Industries: Acute care hospitals (OR, ICU, ED), ambulatory surgical centers, emergency medical services (EMS) systems, and long-term acute care (LTAC) facilities.
-
Specific Operational Use Cases:
-
General anesthesia administration requiring positive pressure ventilation
-
Emergency intubation in respiratory arrest or airway compromise
-
Prolonged mechanical ventilation in ICU (e.g., COVID-19 ARDS, post-cardiac arrest)
-
-
Performance Advantage over Alternatives: Unlike low-volume high-pressure cuffs or uncuffed tubes, the HVLP design maintains a reliable seal at 20–30 cm H₂O cuff pressure, dramatically reducing the incidence of tracheal stenosis and aspiration pneumonia. The 8.0 mm size offers optimal airflow resistance (≤2.2 cm H₂O at 30 L/min) for adult male and most female patients.
-
Efficiency/Cost Advantage: Reduced need for cuff pressure monitoring interventions (compared to standard cuffs) lowers nursing workload; decreased post-extubation complications translates to shorter ICU length of stay—a direct system cost reduction of an estimated 2,500–5,000 per major airway complication avoided.
4. Competitive Advantages
-
Quality Consistency: ISO 13485:2016 certified manufacturing; 100% cuff leak and inflation integrity testing prior to packaging; lot traceability to raw material batch.
-
Supply Reliability: Dual-source raw material agreements; safety stock buffer of 3 months’ average demand; 98% on-time-in-full (OTIF) delivery track record for contracted buyers.
-
Logistics Capability: Global distribution hubs (North America, EU, SE Asia) enable 5–7 day express B2B delivery; ambient temperature shipment (no cold chain required).
-
Price Competitiveness: Volume-based tiered pricing with no minimum annual commitment penalty; benchmarked 12–18% below incumbent premium brands for equivalent clinical performance.
-
Environmental & Documentation Support: Recyclable outer packaging (FSC-certified cardboard); full technical file access (CE MDR or FDA 510(k) pre-market notification provided upon NDA); multilingual instructions for use (IFU) and lot-specific Certificate of Analysis (CoA).
5. Commercial & Supply Information
-
Minimum Order Quantity (MOQ): BULK 20 metric tons (MT) – equivalent to approximately 400 master cases (20,000 individual tubes) based on standard packing density. Sample quantities (≤50 units) available for clinical evaluation at nominal fee, creditable toward first bulk order.
-
Loading Capacity (per 20’ standard container): 20 MT net product weight – achieved via palletized master cases; total gross weight including pallets and desiccants not exceeding 22.5 MT per container. Full container load (FCL) only; LCL not offered for bulk pricing tier.



Reviews
There are no reviews yet.