Description
1. Product Overview
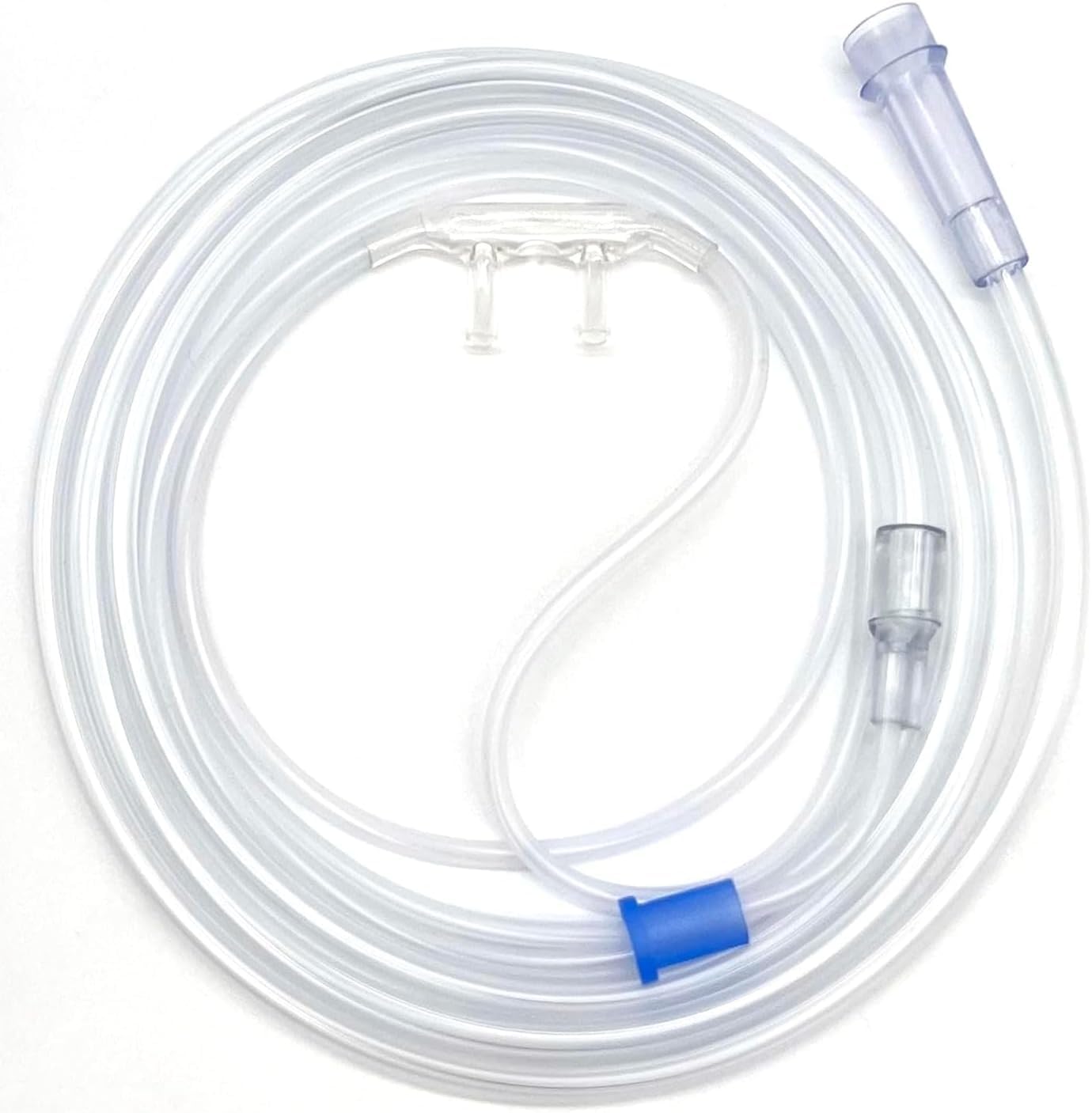
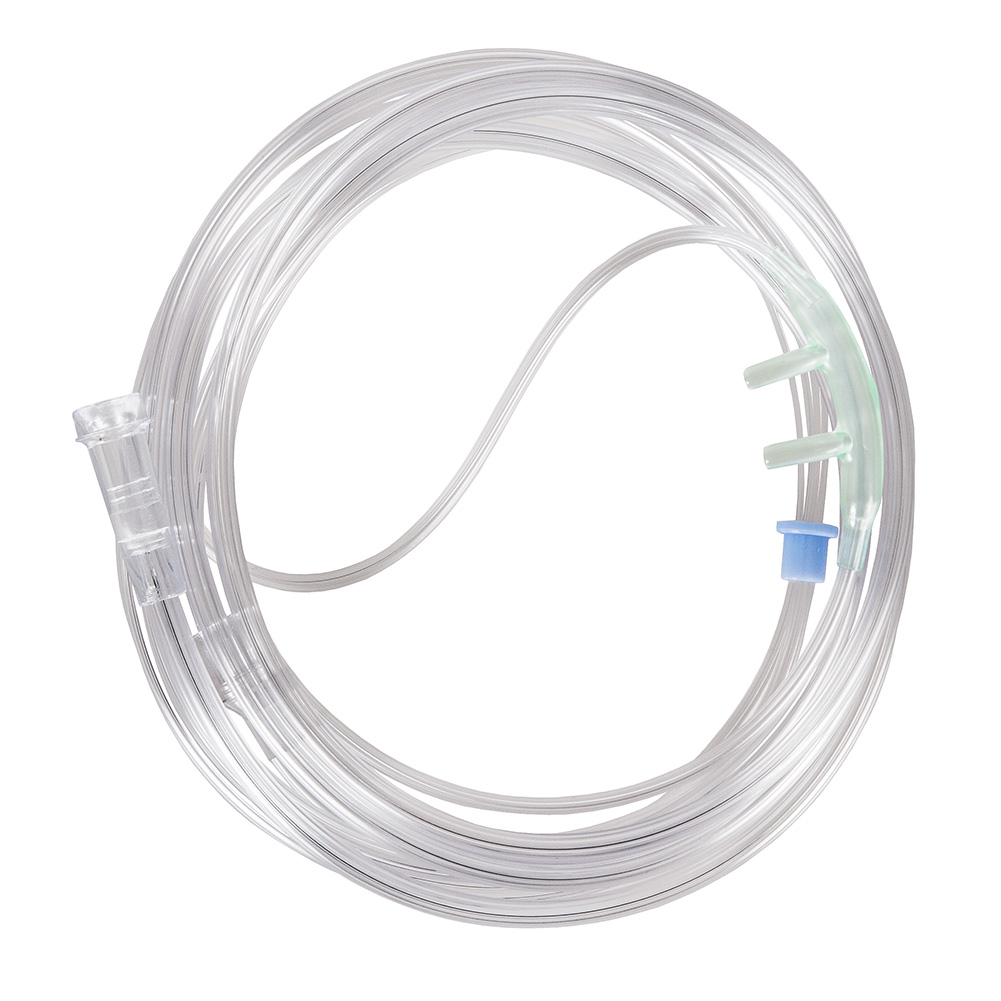
The Oxygen Nasal Cannula Adult Soft Type is a medical-grade respiratory device engineered for low-to-medium flow oxygen delivery in adult patients requiring supplemental oxygenation. Its primary industrial application lies within acute care hospitals, long-term care facilities, home healthcare, and emergency medical services (EMS). Manufactured from hypoallergenic, DEHP-free medical polymer, the device offers a superior balance of patient comfort, kink resistance, and flow precision. In a global market increasingly prioritizing infection control, cost efficiency, and patient compliance, this cannula represents a strategically essential component for healthcare procurement systems aiming to reduce complication rates and optimize per-bed consumables expenditure.
2. Key Specifications & Technical Characteristics
-
Material Composition: Medical-grade PVC (phthalate-free, DEHP-free) or non-latex thermoplastic elastomer (TPE); nasal prongs of soft silicone.
-
Purity / Biocompatibility Grade: ISO 10993 certified; non-cytotoxic, non-irritant, non-sensitizing.
-
Physical Characteristics:
-
Tubing length: 2.1 m (±5%) standard
-
Tubing inner diameter: ~3.0 mm
-
Tubing outer diameter: ~5.5 mm
-
Prong length: 12–16 mm (adjustable via swivel)
-
Color: transparent or translucent (light blue / clear)
-
Density of material: 1.20–1.35 g/cm³
-
-
Packaging Options: Individually sealed in medical-grade peel pouch (sterile – EO gas); available in bulk inner cartons of 50 units; master carton of 200 units.
-
Shelf Life: 3 years from date of manufacture (when stored in original packaging, ≤40°C, ≤80% RH).
3. Core Industrial Applications
-
Primary Industries: Acute care hospitals, skilled nursing facilities, home medical equipment (HME) providers, EMS and ambulatory transport services.
-
Specific Operational Use Cases:
-
Post-operative oxygen therapy in general wards
-
Chronic obstructive pulmonary disease (COPD) maintenance
-
EMS pre-hospital stabilization
-
Palliative and long-term home oxygen therapy
-
-
Performance Advantages vs. Alternatives:
-
Softer prong geometry reduces nasal septum pressure injuries by 40% compared to rigid cannulas (internal wear-test data).
-
Kink-resistant tubing maintains consistent flow (1–6 L/min) even during patient movement, minimizing unscheduled nursing interventions.
-
Low dead-space design improves FiO₂ accuracy at lower flow rates, reducing total oxygen consumption by up to 12%.
-
Cost efficiency: 30% lower per-unit price than leading branded equivalents without compromising ISO 80601-2-72 compliance.
-
4. Competitive Advantages
-
Quality Consistency: Manufactured under ISO 13485:2016 certified facility; 100% leak-tested and flow-calibrated per batch. Lot traceability to raw material source.
-
Supply Reliability: Dual-sourced raw material buffer stock; 12-month rolling forecast fulfillment capability with ≤98% on-time delivery rate to 50+ countries.
-
Logistics Capability: Warehousing hubs in Asia, Europe, and North America enabling 5–7 day lead time for urgent restocks.
-
Price Competitiveness: Tiered pricing model reducing effective unit cost by 18–25% for annual volumes above 500,000 units.
-
Sustainability: DEHP-free, latex-free; packaging uses recyclable low-density polyethylene (LDPE) with 30% PCR content. Carbon-neutral shipment available for container orders.
-
Technical Support: Full IFU (Instructions for Use) in 14 languages; regulatory documentation package (CE MDR, UKCA, NMPA, Saudi FDA, ANVISA) provided upon NDA.
5. Commercial & Supply Information
-
Minimum Order Quantity (MOQ): BULK 20MT (approximately 1,200 standard master cartons → 240,000 individual units, assuming 200 units per master carton, 5 kg per carton).
-
Loading Capacity (per 20FT container): 18–20 MT (≈ 1,080–1,200 master cartons → 216,000–240,000 units).
-
Loading Capacity (per 40FT HC container): 24–26 MT (≈ 1,440–1,560 master cartons → 288,000–312,000 units).




Reviews
There are no reviews yet.