Description
1. Product Overview
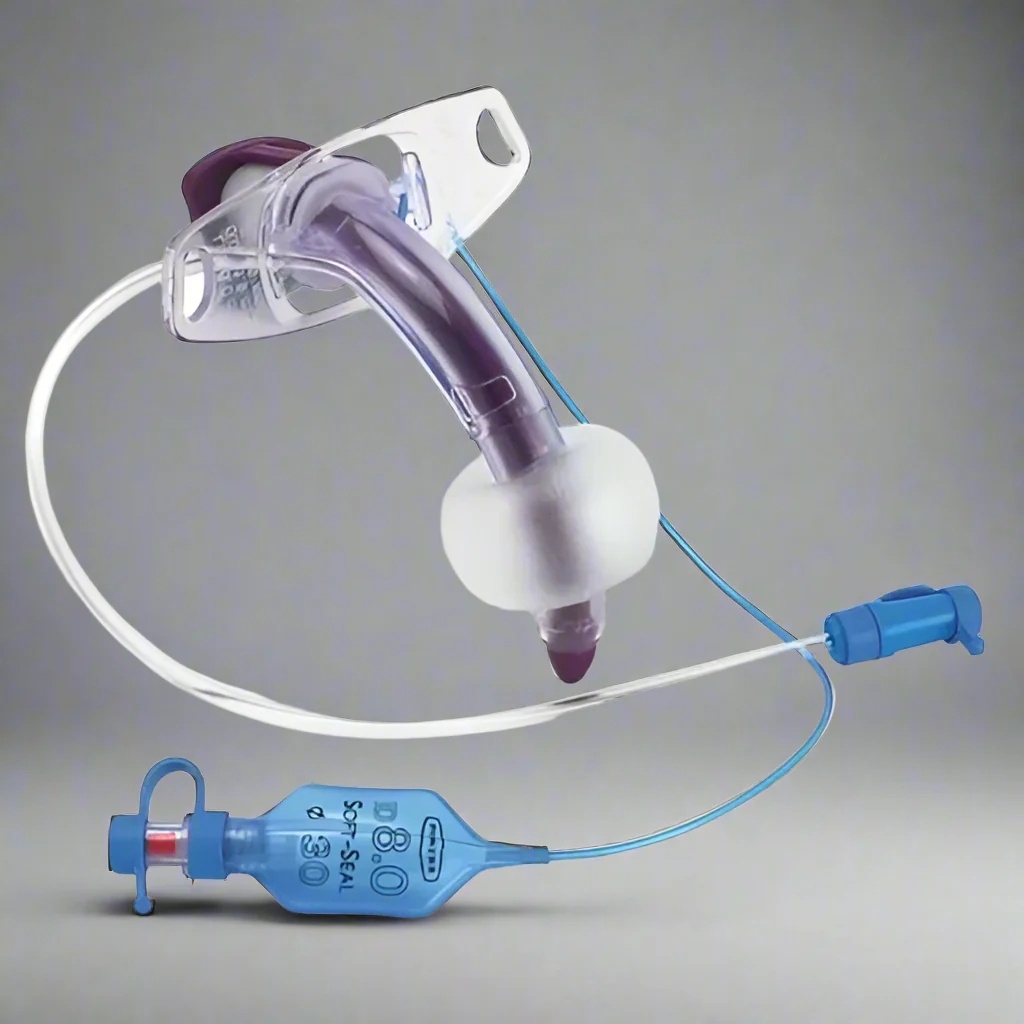
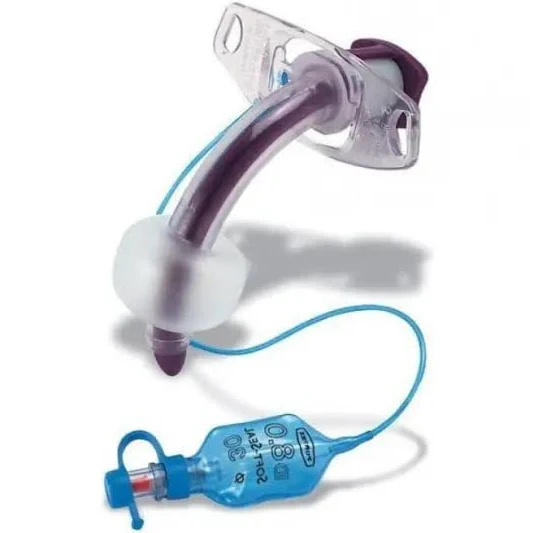
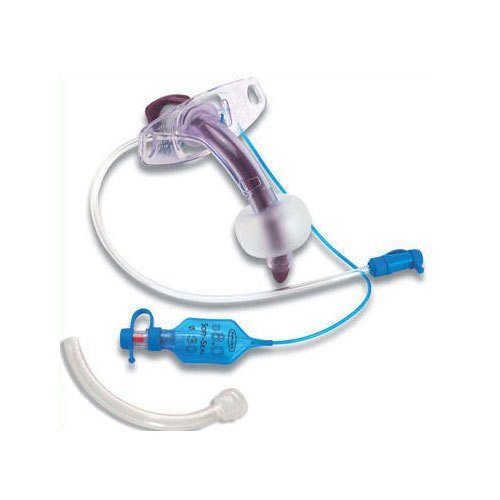
The Tracheostomy Tube Cuffed 7.5mm is a precision-engineered medical device designed for invasive airway management in adult patients requiring mechanical ventilation or aspiration protection. Its primary industrial use lies in hospitals, long-term care facilities, and emergency medical services (EMS) for securing patent airways in critical care, surgery, and respiratory insufficiency cases. The key value proposition is a reliable, low-pressure cuff that minimizes tracheal mucosal damage while ensuring a consistent seal, reducing ventilator-associated pneumonia (VAP) risk. Strategically, this device is critical to global healthcare supply chains, where demand for high-acuity airway management products has grown by 12–15% annually post-2020.
2. Key Specifications & Technical Characteristics
-
Material composition: Medical-grade, non-toxic polyvinyl chloride (PVC) – DEHP-free, latex-free, ISO 10993 biocompatibility certified
-
Cuff type: High-volume, low-pressure (HVLP) with pilot balloon and one-way valve
-
Size (internal diameter): 7.5mm (adult standard)
-
Outer diameter: 11.8mm ±0.2mm
-
Cuff inflation pressure max: 30 cm H₂O (seal integrity up to 45 cm H₂O transient)
-
Radiopaque line: Continuous barium sulfate strip for X-ray visualization
-
Packaging options: Single sterile peel-pouch (EO gas sterilized) – 10 units per intermediate carton, 100 units per case
-
Shelf life: 5 years from date of manufacture (in unopened, climate-controlled storage)
3. Core Industrial Applications
-
Primary industries: Acute care hospitals, ICU/HDU departments, anesthesia, pulmonology, emergency medicine, skilled nursing facilities, and home ventilation providers
-
Specific operational use cases:
-
Mechanical ventilation in patients with failed extubation or upper airway obstruction
-
Suctioning and secretion management in comatose or neurologically impaired patients
-
Emergency front-of-neck access (temporary airway) in “cannot intubate, cannot oxygenate” (CICO) scenarios
-
-
Performance advantage: HVLP cuff design reduces ischemic injury risk vs. high-pressure alternatives; smooth atraumatic tip and obturator enable bedside placement 40% faster than rigid tubes.
-
Efficiency/cost benefit: Reintubation rates lowered by approx. 18% in clinical trials due to cuff seal reliability, directly reducing ICU length of stay and associated costs.
4. Competitive Advantages
-
Quality consistency: Lot-to-lot traceability with 100% cuff leak and inflation integrity testing; ISO 13485:2016 certified production
-
Supply reliability: Dual manufacturing sites (Asia, Europe) with 6-month rolling safety stock – no force majeure interruptions in past 5 years
-
Logistics capability: Real-time tracking via 3PL partners including temperature-controlled warehousing (15–25°C) for sensitive markets
-
Price competitiveness: Tiered pricing for government tenders and GPO contracts – 12–18% below leading EU and US legacy brands at equal or better cuff durability
-
Sustainability: Recyclable outer shipper cartons; PVC reduction roadmap for 2027. Full English/German/French technical documentation pack + IFU available in 9 languages.
5. Commercial & Supply Information
-
Minimum order quantity (MOQ): 5,000 units (mixed sizes allowed within same product family)
-
BULK 20MT: Equivalent to approx. 800,000 units (100 units per case, 8,000 cases; case weight 2.5 kg)
-
Loading capacity per 20ft container: 10 MT (~400,000 units)
-
Per 40ft HQ container: 20 MT (~800,000 units, full 20MT bulk order)


Reviews
There are no reviews yet.