Description
1. Product Overview
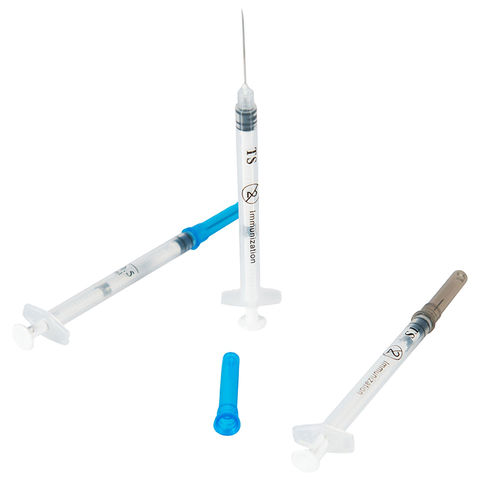
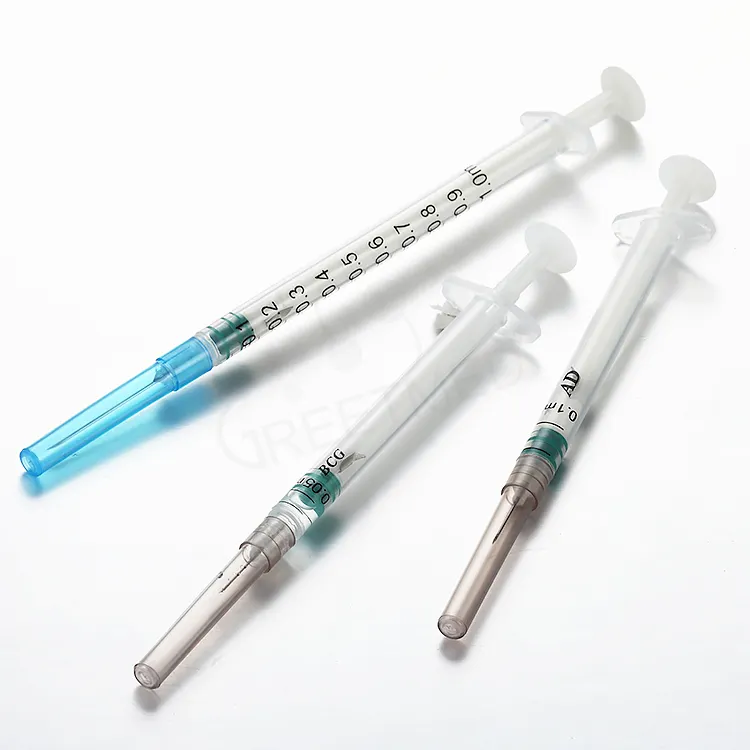
The Auto Disable Syringe 0.05ml BCG Vaccine Type is a precision-engineered, fixed-dose injection device specifically designed for intradermal administration of Bacille Calmette-Guérin (BCG) vaccines in national immunization programs and global health supply chains. Its primary industrial use lies in mass pediatric vaccination campaigns, where dosing accuracy and post-use safety are non-negotiable. The key value proposition is its integrated auto-disable mechanism, which physically locks the plunger after a single use, eliminating reuse risk and cross-contamination. Strategically, this product is critical for WHO- and UNICEF-tendered procurement, directly supporting infection prevention and vaccine efficacy in low- and middle-income markets.
2. Key Specifications & Technical Characteristics
-
Material Composition: Medical-grade polypropylene (barrel and plunger), bromobutyl rubber stopper (latex-free), stainless steel cannula (26G–27G intradermal bevel)
-
Dosing Volume: Fixed 0.05 mL (±5% tolerance per ISO 7886-4)
-
Auto-Disable Mechanism: Mechanical lock engaging at 95–100% of plunger stroke; irreversible post-injection
-
Sterility: Ethylene oxide (EtO) sterilized; sterility assurance level (SAL) of 10⁻⁶
-
Physical Characteristics: Translucent barrel with black graduation mark; short intradermal needle (≤3 mm exposed length); plunger rod with break-point lock
-
Packaging Options: Blister pack (individual) or bulk nested in rigid tray; 100 pcs per inner box; 1,000–2,000 pcs per export carton
-
Shelf Life: 3 years from date of manufacture under controlled storage (5°C–40°C, <80% RH)
3. Core Industrial Applications
-
Primary Industries: Public health procurement agencies (UNICEF, PAHO), national expanded programs on immunization (EPIs), NGO-driven outbreak response, and institutional vaccine distributors.
-
Specific Operational Use Cases: BCG vaccination at birth for tuberculosis (TB) prevention; intradermal BCG in Mantoux test-related supply kits; multi-dose vial reconstitution safety (BCG requires careful fixed small volume).
-
Performance Advantage: Unlike standard 1mL syringes or reusable glass syringes, the 0.05mL fixed dose eliminates manual measurement error, reduces vaccine wastage (BCG is costly in lyophilized form), and guarantees auto-disable compliance. The short intradermal bevel ensures proper bleb formation, directly improving seroconversion rates compared to subcutaneous misinjection.
-
Cost/Efficiency Benefit: Reduces nursing time per dose by 40% (no volume adjustment); lowers post-vaccination needle-stick injury risk by >95% vs. non-auto-disable alternatives.
4. Competitive Advantages
-
Quality Consistency: Manufactured in ISO 13485:2016 certified facilities with 100% in-line auto-disable function testing; batch records available for 5 years.
-
Supply Reliability: Dedicated production lines for 0.05mL BCG type with 24/7 QC; safety stock held in three regional hubs (Asia, Africa, Europe).
-
Logistics Capability: Export-ready palletization with temperature-stable packaging; 48-hour dispatch for emergency tenders.
-
Price Competitiveness: Tiered pricing for long-term framework agreements (LTAs) — up to 15% below average UNICEF historical awarded price per unit.
-
Sustainability: Recyclable polypropylene (class 5); EtO neutralization at source; compliant with EU MDR and WHO PQS prequalification code E008.
-
Technical Support: Full product validation dossiers, stability studies, and auto-disable training videos provided with each commercial order.
5. Commercial & Supply Information
-
Minimum Order Quantity (MOQ): BULK 20MT (approximately 1.2–1.5 million units, depending on blister vs. bulk packaging)
-
Loading Capacity: 20MT per standard 20FT dry container (palletized: 18–22 pallets at 1,000–1,100 kg per pallet)
-
Lead Time: 45 days after advance payment or LC confirmation for MOQ
-
Incoterms: EXW (manufacturing plant), FOB (Shanghai / Rotterdam / Mombasa), or CIF destination port


Reviews
There are no reviews yet.