Description
1. Product Overview
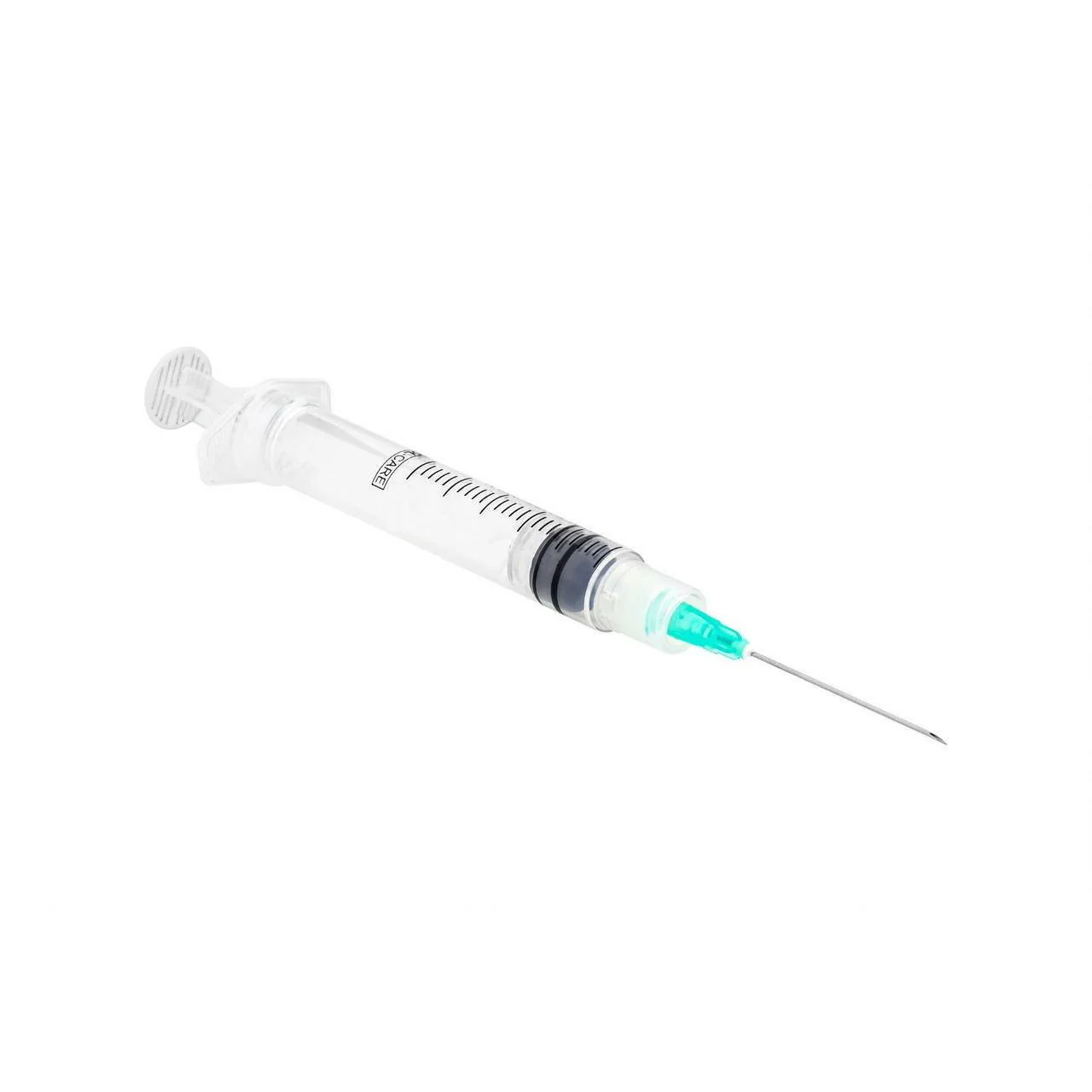
The Hypodermic Needle 23G x 1″ Sterile Disposable is a precision-engineered, single-use medical device designed for routine parenteral administration, fluid aspiration, and intravenous access in clinical and field settings. Its primary industrial use spans hospital networks, diagnostic laboratories, outpatient surgical centers, and large-scale vaccination campaigns. The key value proposition lies in its optimal gauge-to-length ratio (23G x 1″)—balancing patient comfort with efficient flow rates for medium-viscosity solutions—combined with gamma-sterilized, tamper-evident packaging. Strategically, this needle type represents a high-turnover, essential consumable in global healthcare supply chains, where standardization, sterility integrity, and cost-per-unit predictability directly influence procurement ROI and clinical safety metrics.
2. Key Specifications & Technical Characteristics
-
Material Composition: Medical-grade stainless steel (AISI 304/316 equivalent) cannula; polypropylene or ABS luer lock hub; silicone-free lubrication for reduced tissue drag
-
Sterility & Grade: Gamma or EtO sterilized; non-pyrogenic, latex-free, ISO 7864:2016 compliant; RNase/DNase-free upon request
-
Physical Characteristics:
-
Gauge: 23G (outer diameter: 0.641 mm / 0.0252″)
-
Length: 1 inch (25.4 mm)
-
Bevel design: 3-bevel short bevel for precision intradermal/subcutaneous use
-
Color code: Blue (ISO 6009 standard)
-
-
Packaging Options:
-
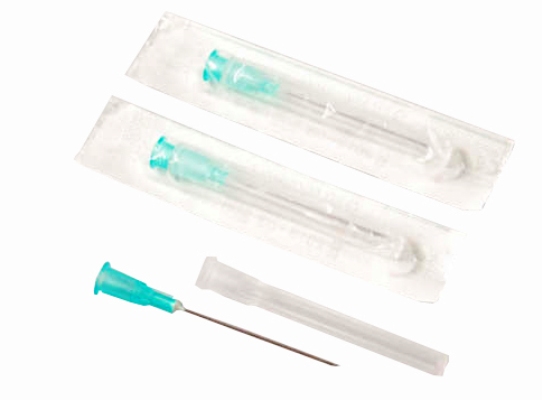
Individual blister peel-pack (medical grade paper/Tyvek & PET film)
-
Bulk: 100pcs/box, 50 boxes/case (5,000pcs/case)
-
Sterile shelf-life: 5 years from date of manufacture in unopened packaging
-
-
Quality Compliance: CE marking (MDR Class IIa), FDA 510(k) cleared (upon applicable lot), ISO 13485 certified production
3. Core Industrial Applications
-
Primary Industries: Hospital group purchasing organizations (GPOs), clinical research organizations (CROs), public health authorities, veterinary clinics, pharmaceutical fill-finish lines (for QC sampling)
-
Specific Operational Use Cases:
-
Subcutaneous or intramuscular vaccine delivery (e.g., influenza, hepatitis B)
-
Aspiration of small-volume fluid samples from drug vials or biological reservoirs
-
Preparation of infusion admixtures in hospital pharmacies
-
Routine blood draws in low-volume diagnostics (preferred for fragile veins in pediatrics/geriatrics)
-
-
Performance Advantage over Alternatives:
-
Compared to 25G: 23G offers 40% faster flow rate for suspensions or higher-viscosity injectables, reducing plunger force and clinician hand fatigue.
-
Compared to 21G: Reduced patient pain response and lower risk of coring in rubber stoppers.
-
Sterility barrier packaging maintains integrity through -20°C to +50°C supply chains, outperforming low-cost competitors whose seals fail under humidity variation.
-
4. Competitive Advantages
-
Quality Consistency: Lot-to-lot cannula sharpness measured via penetration force testing (<0.8N per ISO 7864); each production batch includes burr-free visual inspection at 5x magnification.
-
Supply Reliability: Dual manufacturing locations (Asia + Europe) with buffer safety stock of 50M units; 98% on-time-in-full (OTIF) delivery performance over 12 quarters.
-
Logistics Capability: Pre-collated master cartons optimized for automated dispensing in high-volume vaccination sites; RFID-ready case labeling for inventory management integration.
-
Price Competitiveness: Tiered pricing achieves 12–18% below leading brand (BD, Nipro) while maintaining equivalent or superior penetration force metrics.
-
Sustainability: Recyclable polypropylene hubs; packaging reduced by 22% vs industry average; available carbon-neutral shipping on full container loads.
-
Technical Support: Full declaration of conformity, IFU with 15 languages, and regulatory documentation package (Eudamed, UDI) available for procurement audits within 24 hours.
5. Commercial & Supply Information
-
Minimum Order Quantity (MOQ): BULK – 50,000 units (10 cases) for first trial order; subsequent orders: 1,000,000 units/year commitments eligible for direct mill pricing.
-
Loading Capacity (MT per container):
-
20MT total per standard 40ft HC container.
-
Equivalent to ~1,250,000 units (case dimensions: 48 x 32 x 38 cm; 12kg per 5,000-unit case; 166 cases per container net, no pallet overhang).
-
Half-container loads (10MT) available at incremental logistics charge.
-


Reviews
There are no reviews yet.