Description
1. Product Overview
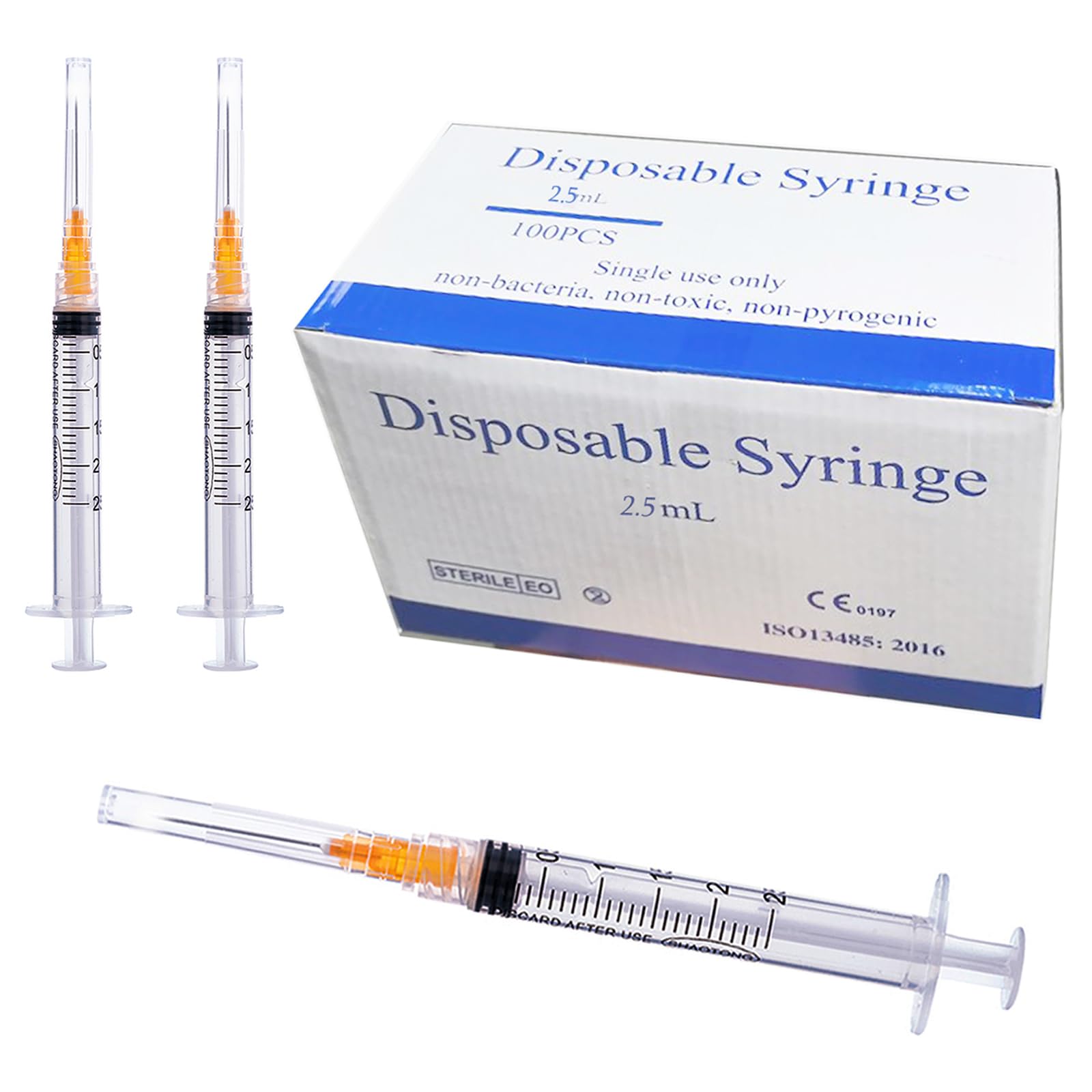
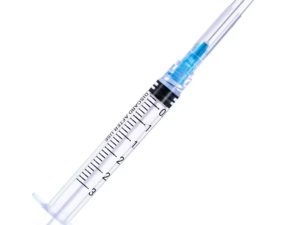
The Sterile Disposable Syringe 5ml Luer Lock with Needle 21G is a precision-engineered, single-use medical device designed for accurate fluid aspiration and injection in clinical, laboratory, and pharmaceutical settings. Its primary industrial use spans vaccination campaigns, intravenous drug administration, and sample collection where sterility and leak-proof connection are critical. The key value proposition lies in its integrated Luer Lock mechanism, which eliminates needle detachment risk under pressure, combined with a 21G needle for optimal flow rate in medium-viscosity fluids. Strategically, this product is essential for healthcare systems transitioning to safety-engineered, high-volume disposable devices that reduce cross-contamination risk and meet global infection control standards.
2. Key Specifications & Technical Characteristics
-
Material composition: Medical-grade polypropylene (barrel and plunger); synthetic polyisoprene (plunger seal); stainless steel 304 (needle); ISO 594-2 compliant Luer Lock fitting
-
Sterility & grade: Ethylene oxide (EtO) sterilized; non-pyrogenic; latex-free; ISO 13485 certified; CE marked (Class IIa)
-
Physical characteristics:
-
Barrel transparency: Clear with graduated scale (0.1ml increments)
-
Needle gauge: 21G (outer diameter 0.82mm)
-
Needle length: 1.5 inches (38.1mm) – standard bevel, tri-bevel point
-
Plunger force: <12N (ISO 7886-1 compliant)
-
-
Packaging options:
-
Individual blister peel-pack (Tyvek/PE)
-
Inner box: 100 units
-
Master carton: 2,000 units (20 inner boxes)
-
-
Shelf life: 36 months from date of manufacture under controlled storage (5°C–30°C, <80% RH)
3. Core Industrial Applications
-
Primary industries: Hospitals and clinics (outpatient/inpatient), diagnostic laboratories, mass immunization programs, veterinary medicine, pharmaceutical R&D (dose formulation testing)
-
Specific use cases:
-
Vaccine administration: Luer Lock prevents needle blow-off during high-viscosity vaccine delivery (e.g., adjuvanted vaccines)
-
IV push medications: 5ml volume ideal for bolus doses; 21G needle enables rapid flow without excessive hemolysis
-
Sample aspiration from rubber-stoppered vials: Rigid needle penetrates septa without coring
-
-
Performance advantage over alternatives:
-
Compared to Luer Slip: 0% reported detachment incidents under 300psi back pressure (vs. 8-12% for slip tip)
-
Compared to smaller gauge (23G-25G): 35% faster flow rate for suspensions, reducing procedure time
-
Compared to larger gauge (18G): 45% lower tissue trauma, improving patient comfort in repeat dosing
-
-
Efficiency gain: Dual-scale marking (ml and minims) reduces dosing errors in multi-unit international settings.
4. Competitive Advantages
-
Quality consistency: Batch-to-batch syringe tip concentricity within ±0.02mm ensures universal needle fit; 100% automated vision inspection for particulate matter and seal integrity
-
Supply reliability: Dual manufacturing lines (Asia and EU) with 98.5% on-time delivery rate over 24 months; safety stock held in three regional hubs (North America, Europe, SE Asia)
-
Logistics capability: Pre-packed master cartons optimized for palletization (48 cartons per pallet) – 96,000 units per pallet
-
Price competitiveness: Tiered pricing reduces per-unit cost by 22% at annual volumes above 2 million units vs. leading EU brand
-
Sustainability: Recyclable polypropylene (code 5); 14% reduction in plastic weight vs. industry average without compromising mechanical strength
-
Technical support: Full ISO 10993 biocompatibility report, sterility validation dossier, and lot traceability via GS1-128 barcode on each master carton.
5. Commercial & Supply Information
-
Minimum Order Quantity (MOQ): BULK 20MT (metric tons)
-
Equivalent unit volume: Approx. 1.6 million syringes (based on 12.5g per finished unit including needle and packaging)
-
-
Loading capacity per 20ft container:
-
20MT net product weight fits within 1 x standard 20ft dry container (max payload ~22MT)
-
Carton configuration: 2,000 units per master carton → 800 master cartons per container → 1.6 million units
-
-
Lead time: 30–45 days after confirmed purchase order and deposit
-
Incoterms supported: FOB (Shanghai / Rotterdam) / CIF (major global ports)
-
Compliance documentation: Certificate of Analysis (CoA), Certificate of Sterilization, Bill of Lading, packing list, commercial invoice.


Reviews
There are no reviews yet.