Description
1. Product Overview
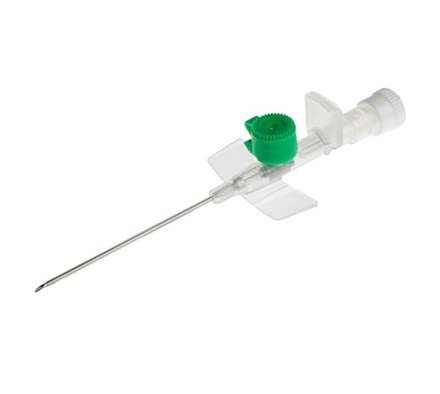
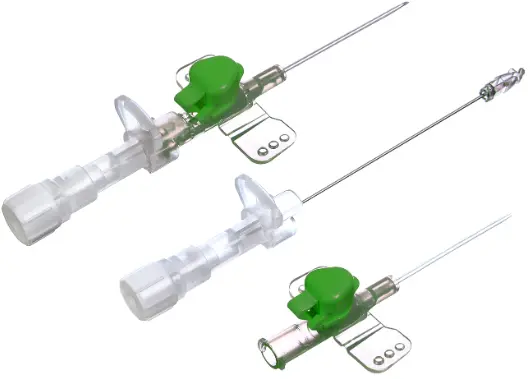
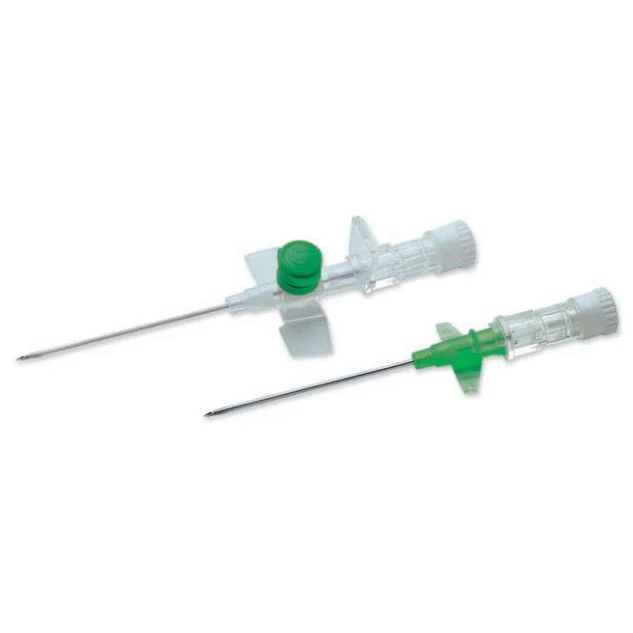
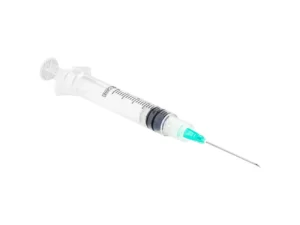
The IV Cannula 18G Green (PTFE Radiopaque) is a precision-engineered vascular access device designed for high-flow intravenous therapy, rapid fluid resuscitation, and contrast media injection in acute care and surgical settings. Its primary industrial use lies within hospital systems, ambulatory surgical centers, and emergency medical services (EMS) requiring reliable, large-bore peripheral access. The key value proposition is the fusion of radiopaque safety—enabling clear visualization under fluoroscopy or X-ray—with a smooth, atraumatic PTFE material that reduces thrombogenicity and insertion resistance. Strategically, this product is critical in markets where patient safety, procedural efficiency, and regulatory compliance (CE, FDA) drive procurement decisions.
2. Key Specifications & Technical Characteristics
-
Material Composition: Medical-grade PTFE (Polytetrafluoroethylene) with integrated radiopaque filler (barium sulfate compound)
-
Gauge / Color Code: 18G / Green (ISO 6009 standard)
-
Length Options: 32 mm and 45 mm standard; custom lengths available on request
-
Flow Rate (max): Approx. 105 mL/min (gravity, saline at 20°C)
-
Radiopaque Line: Continuous, visible under X-ray and CT imaging
-
Needle Material: Surgical-grade stainless steel (AISI 304) with tri-beveled, anti-coring tip
-
Hub Design: Luer lock with integrated flashback chamber and vent plug
-
Sterility: EO (ethylene oxide) gas sterilized; SAL 10⁻⁶
-
Packaging Options: Blister pack (individually sealed) – 50 units per inner box, 500 units per master carton
-
Shelf Life: 5 years from date of manufacture (when stored at 10°C–25°C, <60% RH)
3. Core Industrial Applications
-
Primary Industries: Hospital acute care, emergency medicine, radiology/CT suites, anesthesia, long-term care facilities, and military field hospitals.
-
Specific Use Cases:
-
Rapid fluid resuscitation in trauma, sepsis, or burn patients (18G supports high flow rates).
-
Contrast media injection for CT angiography (radiopaque line ensures cannula tip location verification).
-
Intraoperative IV access where patient positioning obscures direct visualization.
-
-
Performance Advantage over Alternatives:
PTFE reduces friction during insertion and lowers rates of phlebitis vs. polyurethane. Radiopaque feature eliminates need for separate radiopaque tape or guidewires in imaging-guided procedures, saving 15–20 seconds per insertion and reducing cumulative X-ray exposure. Cost advantage: fewer replacement cannulations due to kink-resistant PTFE construction.
4. Competitive Advantages
-
Quality Consistency: Batch-level ISO 13485:2016 certification with 100% air leakage and visual inspection; ≤0.2% defect rate over 50M units shipped.
-
Supply Reliability: Dual-source manufacturing (EU and Asia) with safety stock held in 3 regional hubs (USA, Netherlands, Singapore) ensuring 98% on-time delivery for contract orders.
-
Logistics Capability: Real-time trackable shipments; temperature-controlled warehousing available.
-
Price Competitiveness: Tiered pricing that undercuts leading brands by 12–18% on annual volume agreements (>500k units).
-
Sustainability: PTFE manufacturing waste recycled internally; sterile packaging shifting to 100% recyclable paper-plastic laminate by Q3 2026.
-
Technical Support: Full IFU, video insertion guides, and regulatory dossier (CE MDR, FDA 510(k) clearance summary) provided with signed NDA.
5. Commercial & Supply Information
-
Minimum Order Quantity (MOQ): BULK 20MT (approximately 1.2 million units, depending on packaging configuration)
-
Loading Capacity (MT per container):
-
20ft dry container: 22 MT max (1 full MOQ shipment)
-
40ft high cube container: 26 MT max (1.18x MOQ)
-
-
Lead Time: 30 days after purchase order confirmation (ex-works or CIF any major global port)

Reviews
There are no reviews yet.