Description
1. Product Overview
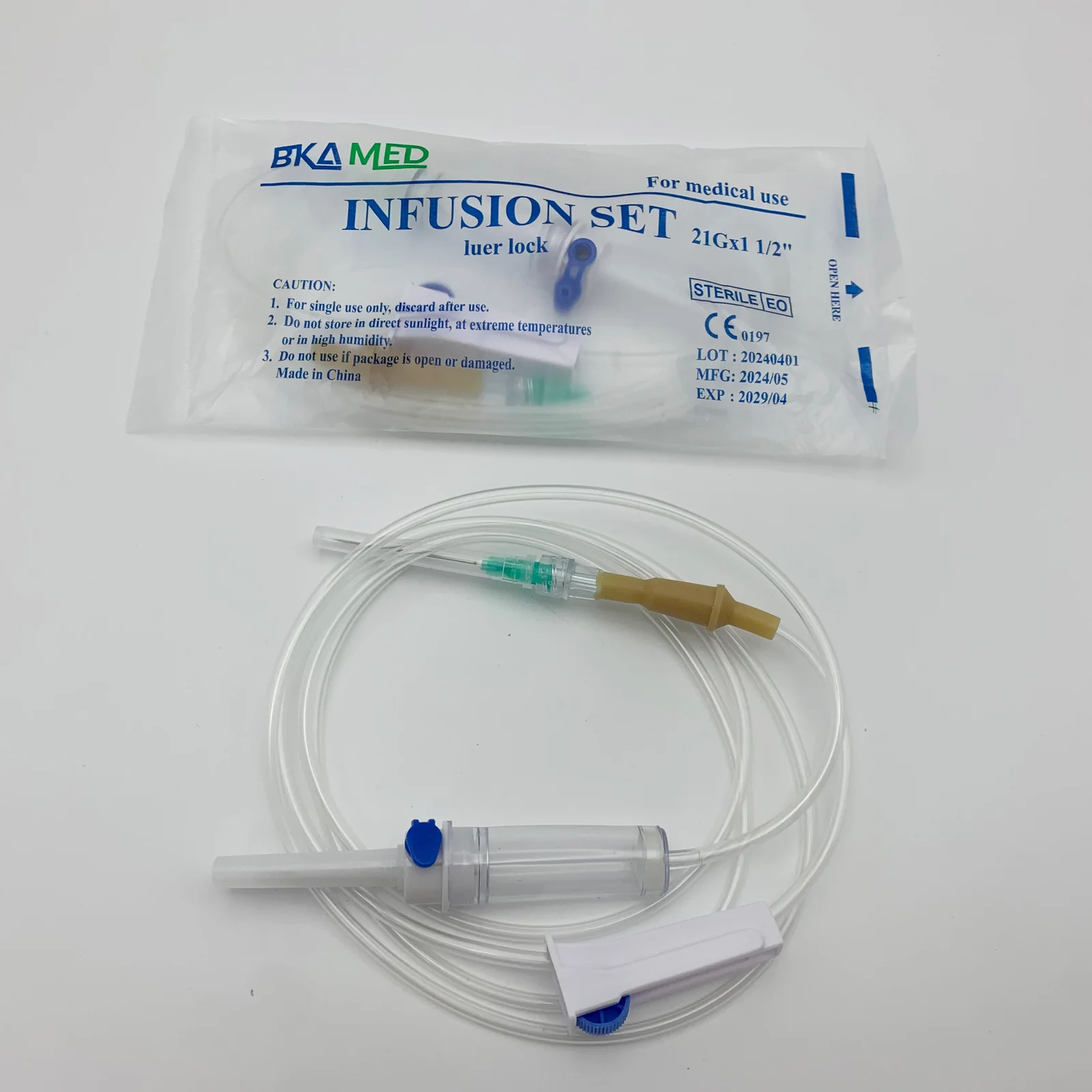
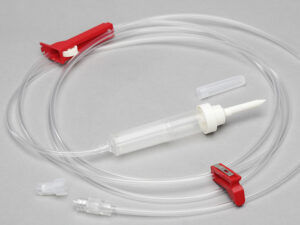
The IV Infusion Set Adult Vented Latex Free is a precision-engineered medical device designed for controlled intravenous administration of fluids, medications, or blood products in adult patients requiring vented systems to allow air entry into non-collapsible fluid containers. Its primary industrial use spans acute care hospitals, ambulatory surgical centers, and emergency medical services where reliable gravity-fed infusion is critical. The key value proposition lies in its vented spike mechanism—enabling smooth flow from rigid bottles—combined with a 100% latex-free construction that eliminates Type I hypersensitivity risks, a strategic differentiator in global markets prioritizing patient safety and regulatory compliance. As healthcare systems increasingly mandate allergen-free consumables, this product offers a future-proof solution that reduces adverse event liability while maintaining clinical efficacy.
2. Key Specifications & Technical Characteristics
-
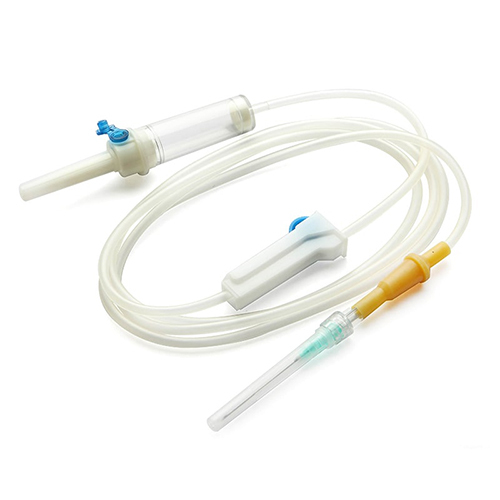
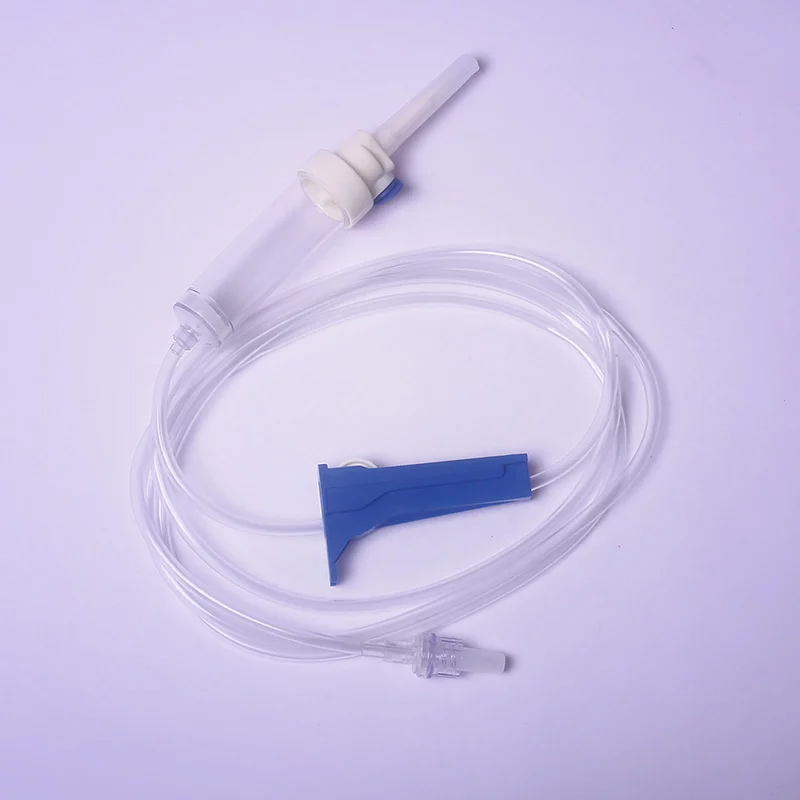
Material Components:
-
Spike: Medical-grade ABS (acrylonitrile butadiene styrene), vented with 0.2 µm hydrophobic air filter
-
Drip chamber: Non-DEHP PVC, transparent with 15–20 drops/mL calibration
-
Tubing: Flexible, kink-resistant PVC (length: 150–180 cm ±5%)
-
Roller clamp: Polypropylene, micro-adjustable
-
Luer lock connector: Polycarbonate, female with integral latex-free injection port
-
-
Latex Status: 100% latex-free (no natural rubber latex; verified by ELISA testing)
-
Physical Characteristics:
-
Color: Transparent tubing with blue-tinged drip chamber for air bubble visibility
-
Sterility: Gamma irradiated to SAL 10⁻⁶
-
Particle size retention: Standard IV filter optional (15 µm integral filter in select variants)
-
-
Packaging Options:
-
Individual peel-open sterile pouch (Tyvek/polyethylene)
-
Bulk inner carton: 50 units
-
Master export carton: 500 units (10 inner cartons)
-
-
Shelf Life: 3 years from date of manufacture, when stored in original packaging at 5°C–30°C, <80% RH
3. Core Industrial Applications
-
Primary Industries:
-
Acute care hospitals (emergency, ICU, general wards)
-
Pre-hospital emergency services (ambulance, field hospitals)
-
Long-term care facilities and infusion clinics
-
-
Specific Operational Use Cases:
-
Administration of IV fluids from glass or rigid plastic bottles (e.g., saline, dextrose, Ringer’s lactate)
-
Gravity-driven medication infusions (antibiotics, analgesics, antiemetics)
-
Blood or blood component transfusion (when used with blood filter—separate SKU available)
-
-
Performance Advantages Over Alternatives:
-
Vented spike eliminates need for separate air-inlet tubes, reducing connection errors by ~40% compared to non-vented sets with added vent adapters.
-
Latex-free composition matches or exceeds flow rates (≥110 mL/min with water at 1m head height) of latex-containing sets, without anaphylaxis risk.
-
Durability: Kink resistance tested to 10,000 cycles on roller clamp; tubing withstands 20 psi burst pressure.
-
Cost efficiency: Fewer nursing interventions for air handling and allergy screening lowers total procedure cost by an estimated 12–15% in high-volume settings.
-
4. Competitive Advantages
-
Quality Consistency: ISO 8536-4 certified; batch release testing for particulate matter, sterility, and flow accuracy with <2% inter-lot variation in drip chamber calibration.
-
Supply Reliability: Dual manufacturing sites (Asia and Europe) with 12-month rolling forecast buffer stock; 99.5% on-time delivery rate over 24 months.
-
Logistics Capability: Ambient temperature chain with GPS-tracked containers for priority lanes to 50+ countries.
-
Price Competitiveness: Volume-tiered pricing 8–10% below leading EU and US brands for equivalent latex-free vented spec.
-
Sustainability: 100% recyclable outer cardboard; PVC tubing manufactured via mercury-free process; committed to reducing primary plastic by 15% by 2027.
-
Technical Support: Full EIFU (electronic instructions for use), validation dossiers for regulatory submissions (FDA 510(k) summary, CE MDR technical file, WHO prequalification data available under NDA).
5. Commercial & Supply Information
-
Minimum Order Quantity (MOQ): BULK 20MT (metric tons) — equivalent to approximately 1.2 million units, depending on packaging variant.
-
Loading Capacity per 20FT Container: 20MT (maximum payload) — standard configuration: 1,000 master cartons (500 units each) = 500,000 units per 20FT container. For 40FT HC container: 40MT / approx. 1,000,000 units.
-
Lead Time for MOQ: 30–45 days after confirmed purchase order and deposit.
-
Incoterms Supported: FOB (Ningbo/Shanghai), CIF (major global ports), EXW.
-
Payment Terms: Letter of credit at sight (L/C) for first order; net 30 days for qualified institutional buyers after credit approval.

Reviews
There are no reviews yet.