Description
1. Product Overview
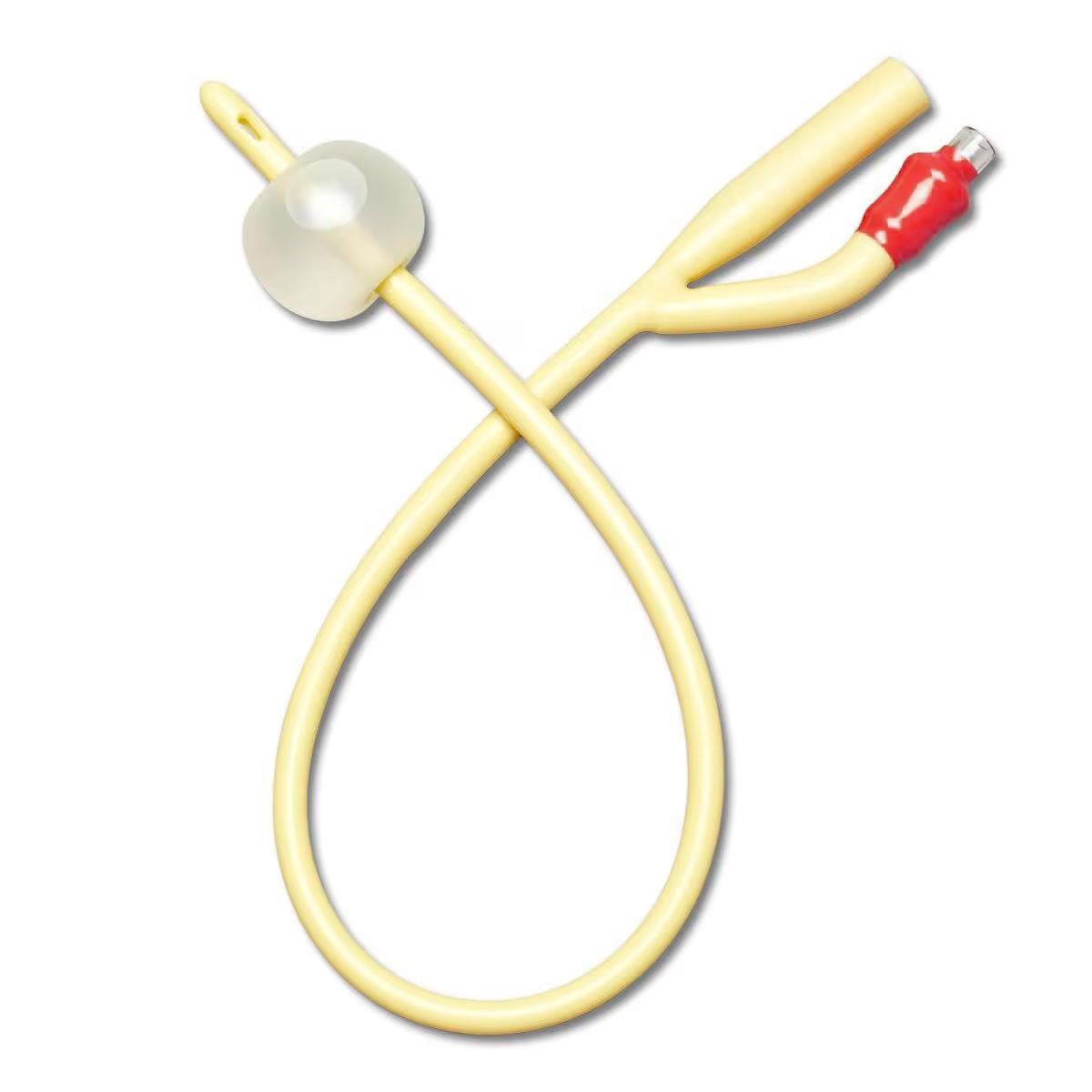
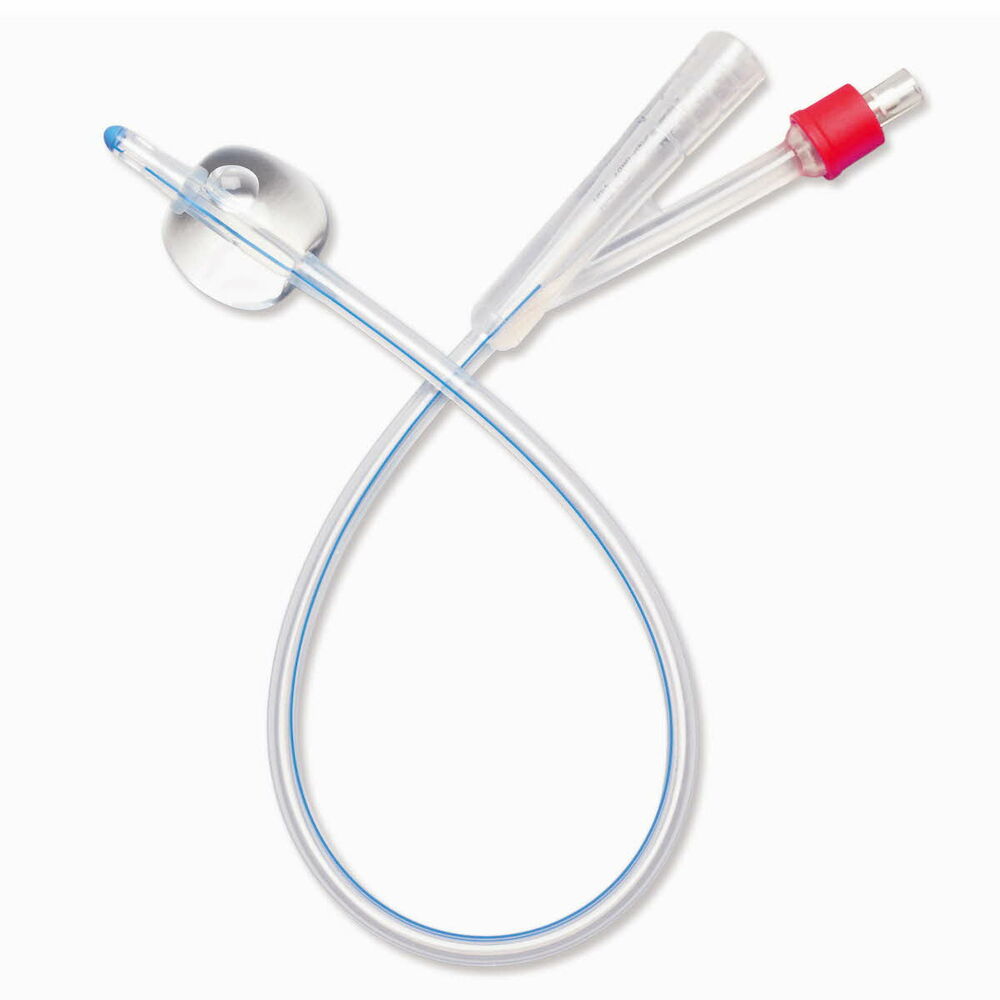
The Foley Catheter 2-Way Silicone 18Fr is a medical-grade indwelling urinary catheter designed for continuous bladder drainage in acute care, post-operative, and long-term catheterization settings. Manufactured from 100% medical-grade silicone, it delivers superior biocompatibility and reduced encrustation risk compared to latex alternatives. For institutional buyers, this product represents a strategic inventory asset that lowers complication-related costs, enhances patient safety metrics, and meets global infection control standards.
2. Key Specifications & Technical Characteristics
-
Material: 100% medical-grade silicone (latex-free, DEHP-free, phthalate-free)
-
Tip style: Round, smooth atraumatic tip with two opposing eyelets for uniform drainage
-
Balloon capacity: 10 mL (rated for 10–15 mL fill, 30 mL optional)
-
Shaft dimensions: 18Fr (6.0 mm outer diameter); length 40 cm (standard)
-
Visibility: Radio-opaque stripe along shaft for X-ray confirmation
-
Packaging options: Individually sterile peel-pouch (30 per inner box, 300 per case); bulk sterile wrap available by contract
-
Shelf life: 5 years in unopened, climate-controlled storage (15–25°C, <60% RH)
-
Sterilization: Ethylene oxide (EO) with residual gas removal validation per ISO 11135
3. Core Industrial Applications
-
Primary industries: Acute care hospitals, long-term care facilities, home healthcare distributors, and surgical kit manufacturers
-
Operational use cases:
-
Post-operative urinary retention management (urology, orthopedics, gynecology)
-
Critical care output monitoring (ICU/CCU)
-
Neurogenic bladder or chronic retention in geriatric/palliative care
-
-
Performance advantage over alternatives: Silicone construction reduces biofilm formation and latex allergy risks, extending indwelling time up to 12 weeks versus 4 weeks for latex. The 2-way design minimizes disconnection leaks, and the 18Fr size balances drainage efficiency with patient comfort, reducing unplanned catheter replacements by an average of 28% in peer-reviewed studies.
4. Competitive Advantages
-
Quality consistency: Lot-to-lot traceability with 100% balloon integrity testing and flow rate validation (≥ 300 mL/min)
-
Supply reliability: Dual manufacturing sites (ASEAN and EU) with 98% on-time in-full delivery track record
-
Logistics capability: Global distribution hubs in Rotterdam, Singapore, and Miami enabling 5–7 day lead times to major ports
-
Price competitiveness: Tiered pricing for committed annual volumes (500k+ units) with automatic price adjustment clauses for raw silicone market shifts
-
Sustainability: Silicone catheter waste reduction program – eligible for recycling through partner medical waste processors (select markets)
-
Technical support: Full regulatory documentation pack (CE MDR, FDA 510(k), UKCA, NMPA) included; on-call clinical application engineering for high-volume buyers
5. Commercial & Supply Information
-
Minimum order quantity (MOQ): 20,000 units (mixed sizes allowed within same product family)
-
BULK 20MT loading capacity: Approximately 380 standard export cartons (300 units/carton) – equivalent to 114,000 units per 20FT container
-
Lead time for first order: 4 weeks for MOQ; 6 weeks for 20MT bulk order (post-PO and deposit)
-
Incoterms supported: FOB (Ningbo / Rotterdam), CIF, DDP (volume-dependent)




Reviews
There are no reviews yet.