Description
1. Product Overview
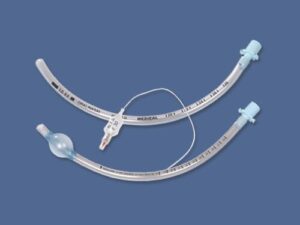
The Endotracheal Tube Cuffed 7.0mm is a Class II medical device engineered for secure airway management in perioperative and critical care settings. Its primary industrial use lies in hospitals, ambulatory surgical centers, and emergency response systems, enabling controlled mechanical ventilation for adult patients requiring intubation. The key value proposition is a reliable, low-pressure cuff seal that minimizes aspiration risk while reducing tracheal mucosal injury—directly improving patient outcomes and reducing ventilator-associated complications. Strategically, this product is critical in global markets facing rising ICU admission rates and surgical volumes, where supply chain consistency and device performance directly impact mortality and reimbursement metrics.
2. Key Specifications & Technical Characteristics
-
Material Composition: Medical-grade, non-toxic polyvinyl chloride (PVC); latex-free, DEHP-free option available upon request
-
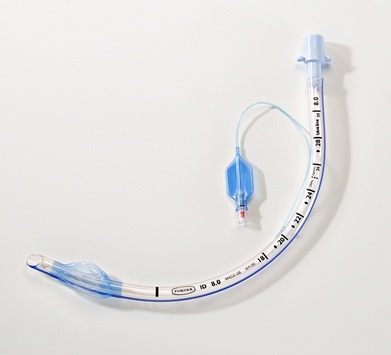
Cuff Type: High-volume, low-pressure (HVLP) compliant cuff with pilot balloon and one-way valve
-
Physical Characteristics:
-
Size: 7.0mm internal diameter
-
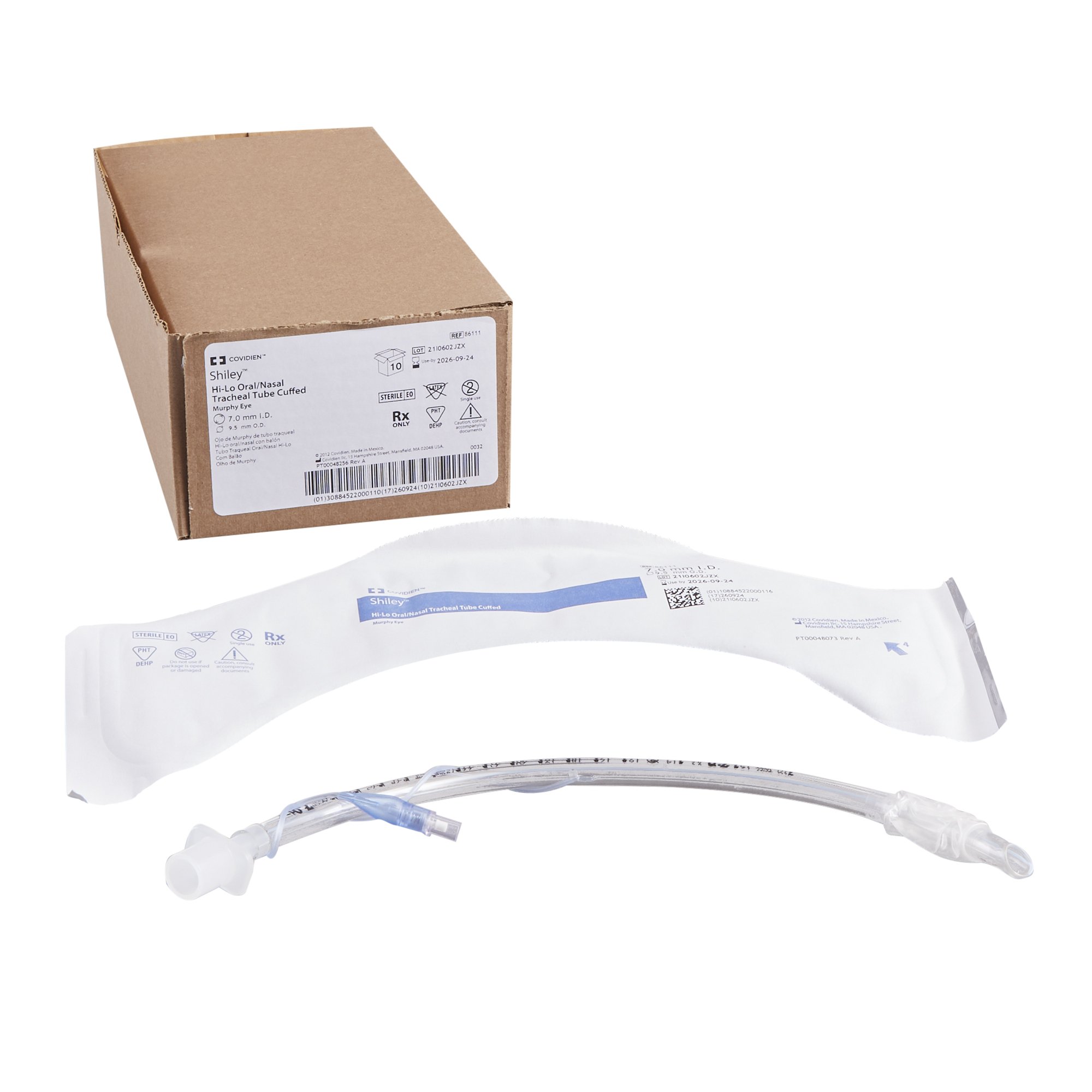
Color: Radiopaque line (blue) for X-ray visualization
-
Form: Preformed (Murphy eye tip) with smooth, atraumatic distal end
-
Depth markings: 1.0cm to 28.0cm from distal tip
-
-
Sterility: Ethylene oxide (EtO) sterilized; sterility assurance level (SAL) 10⁻⁶
-
Packaging Options: Individually sealed peel-open pouch; case pack: 10 units; master carton: 100 units
-
Shelf Life: 5 years from date of manufacture when stored in original, unopened packaging at controlled room temperature (10°C–30°C)
3. Core Industrial Applications
-
Primary Industries: Acute care hospitals, emergency medical services (EMS), military field hospitals, long-term ventilator care facilities
-
Specific Operational Use Cases:
-
General anesthesia during abdominal, thoracic, or head/neck surgery
-
Mechanical ventilation in ICUs for respiratory failure (e.g., COVID-19, pneumonia, COPD)
-
Emergency airway control in pre-hospital or trauma settings
-
-
Performance Advantages:
-
HVLP cuff reduces intracuff pressure drift, lowering post-extubation sore throat (PES) rate by ~30% vs. standard cuffs
-
Radiopaque line ensures rapid confirmation of tube placement via X-ray, reducing malposition risk
-
Murphy eye prevents airway obstruction if the distal opening contacts the tracheal wall
-
-
Cost/Efficiency Benefit: Fewer reintubations and lower rates of ventilator-associated pneumonia (VAP) translate to reduced hospital length of stay and per-patient costs of 4,000–7,000 per avoided VAP episode
4. Competitive Advantages
-
Quality Consistency: ISO 13485:2016 certified manufacturing; 100% cuff leak testing and inflation integrity check prior to packaging
-
Supply Reliability: Dual-sourced raw material inventory with 3 distribution hubs (Asia, EU, NA) guaranteeing <7-day lead time for contract customers
-
Logistics Capability: Real-time batch tracking; cold-chain not required – reduces freight cost and complexity
-
Price Competitiveness: Tiered pricing for annual volumes over 50,000 units offers 15–22% savings over leading EU/US brands at comparable quality
-
Sustainability & Environmental Benefits: Transitioning to recyclable blister packaging (by Q4 2026); PVC recycling program for large hospital groups available
-
Technical Support & Documentation: Full technical file in English, Spanish, and Mandarin; IFU, CE Declaration of Conformity, and FDA 510(k) equivalency documentation available on request
5. Commercial & Supply Information
-
Minimum Order Quantity (MOQ): BULK 20MT (equivalent to approx. 480,000 units of Endotracheal Tube Cuffed 7.0mm, based on 20MT total weight inclusive of master cartons)
-
Loading Capacity (MT per container):
-
20ft dry container: 20 MT maximum (1 full MOQ per container)
-
40ft dry container: 25 MT (partial pallet optimization required for heavy medical devices)
-
*Note for buyers: MOQ defined as 20 metric tons of finished product in mixed-size shipments allowed. Single-size 7.0mm orders require verification with supply chain team.*


Reviews
There are no reviews yet.