Description
1. Product Overview
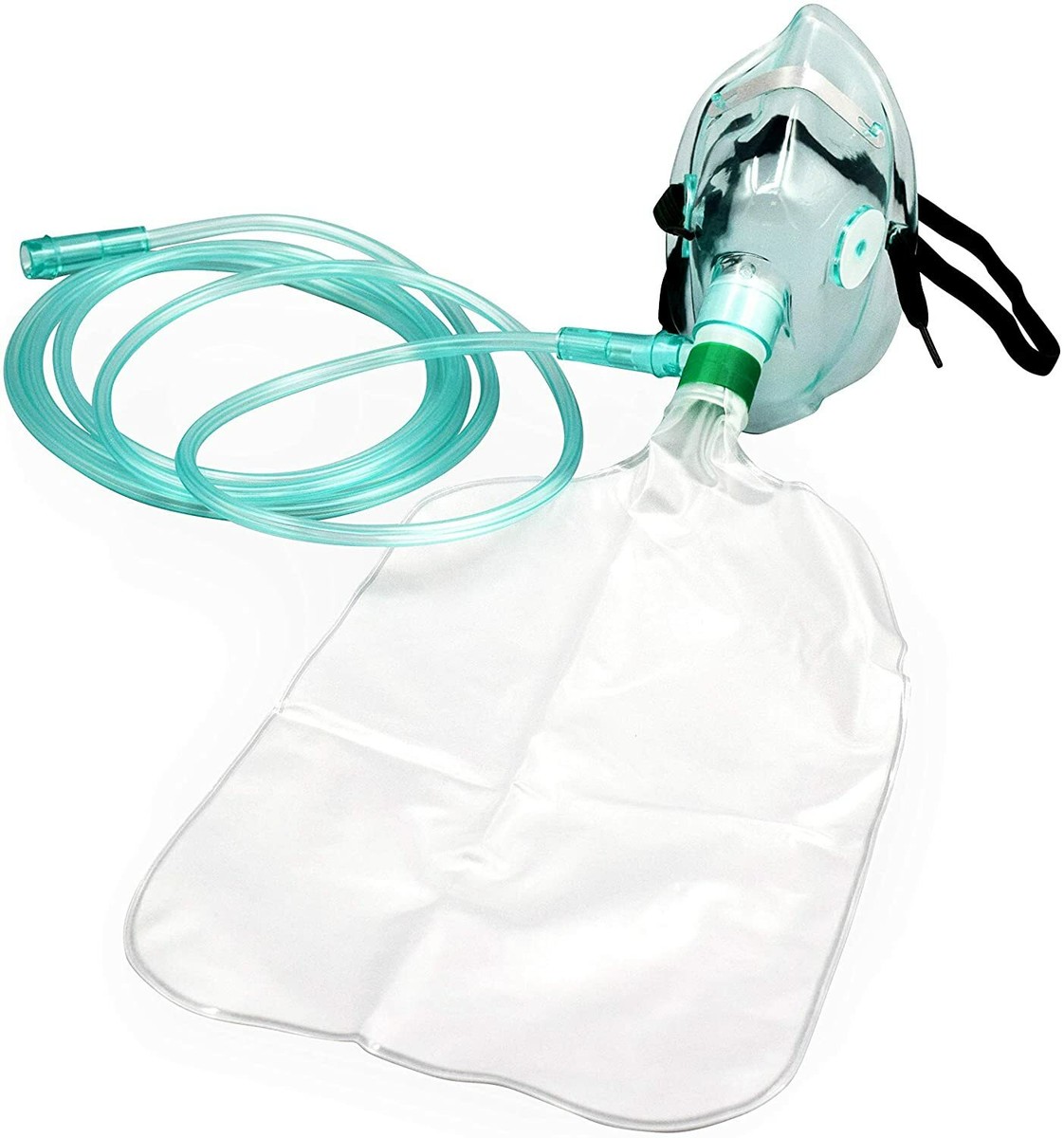
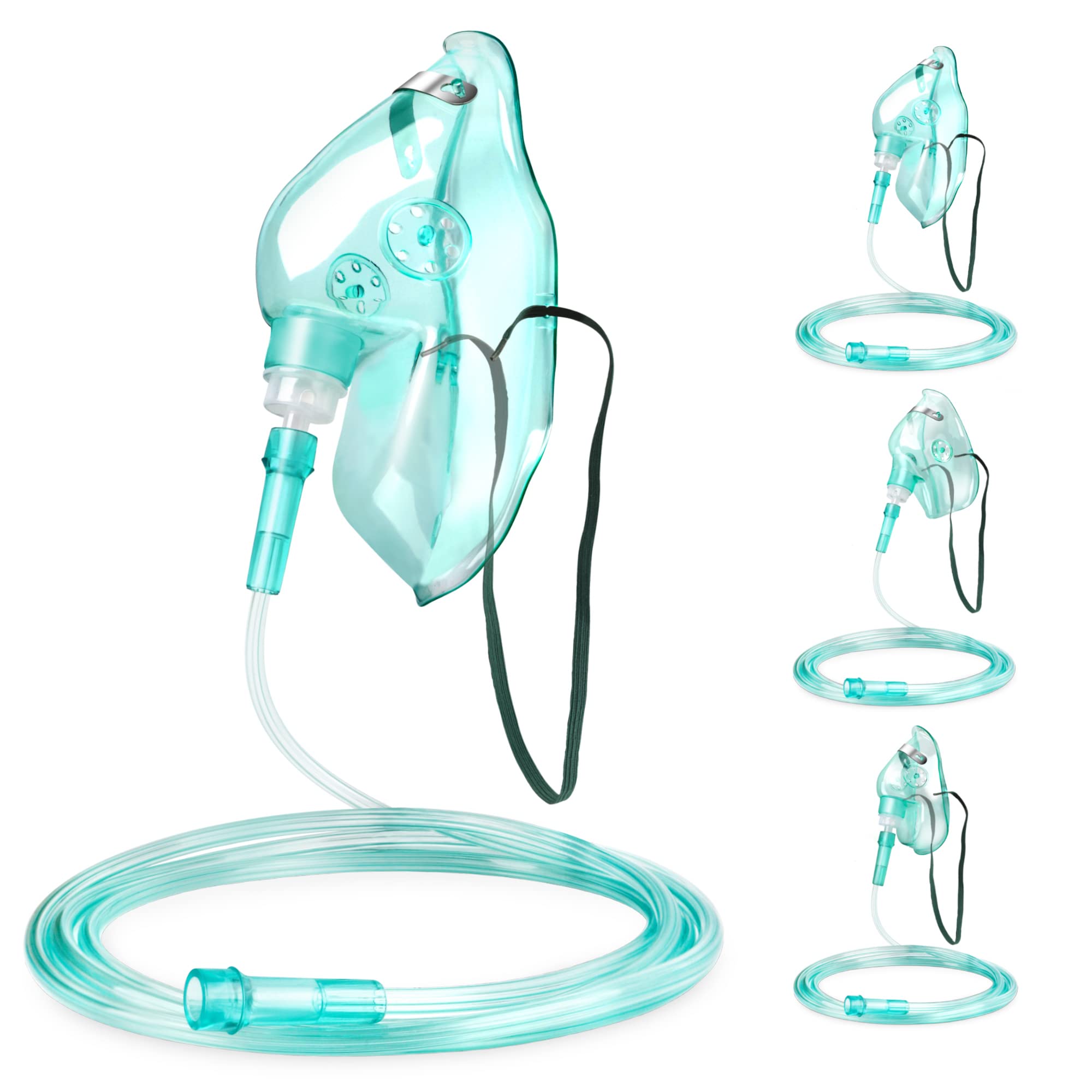
The Venturi Oxygen Mask Adult Complete Set is a precision-engineered respiratory device designed for delivering controlled, high-flow oxygen therapy to adult patients requiring fixed fractional inspired oxygen (FiO₂) concentrations. Its primary industrial use lies in acute care settings—hospitals, long-term care facilities, and emergency medical services—where accurate oxygen titration is critical for conditions such as COPD, pneumonia, and post-operative recovery. The key value proposition is unmatched FiO₂ accuracy (±1–2%) independent of patient breathing patterns, reducing the risk of hypoxemia or hyperoxia. Strategically, this product is vital in global healthcare markets shifting toward value-based, protocol-driven respiratory care, where reducing complications directly impacts patient outcomes and reimbursement.
2. Key Specifications & Technical Characteristics
-
Material components: Medical-grade PVC (phthalate-free), thermoplastic elastomer (TPE) for soft cushion seal, polycarbonate jet nozzle, latex-free elastic strap
-
Purity / grade: ISO 10993 biocompatibility certified, non-pyrogenic, BPA-free
-
Physical characteristics:
-
Transparent mask body for unobstructed monitoring
-
Adult contour fit (nose-to-chin length 9–11 cm)
-
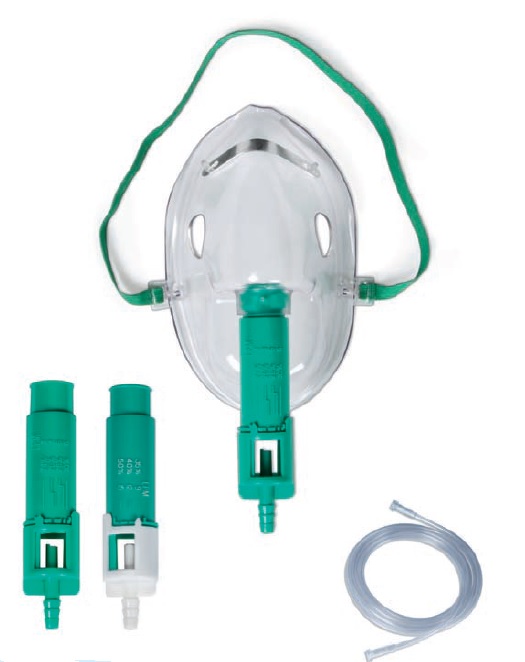
Color-coded Venturi jet adapters (6–8 sizes typically: 24%, 28%, 31%, 35%, 40%, 50%)
-
Anti-occlusion expiratory ports
-
Double-lumen corrugated tubing (length 2.1 m ±5%)
-
-
Packaging options: Individually sealed sterile pouch; 25 sets per inner carton; 4 inner cartons per master case (100 sets)
-
Shelf life: 5 years from manufacture in unopened, climate-controlled storage (15–25°C, <65% RH)
3. Core Industrial Applications
-
Primary industries: Acute care hospitals (ICU, ED, general wards), post-acute skilled nursing facilities, home respiratory therapy providers, disaster preparedness medical stockpiles
-
Specific use cases:
-
COPD patients requiring precise low-flow oxygen (e.g., 24–28% FiO₂) to avoid CO₂ retention
-
Post-anesthesia recovery where variable respiratory rates demand fixed FiO₂
-
Respiratory distress in COVID-19 or pneumonia protocols
-
-
Why this product performs better:
-
Unlike simple masks or nasal cannula, Venturi design entrains a fixed ratio of room air, eliminating FiO₂ variability caused by tachypnea or bradypnea.
-
Color-coded adapters reduce clinician setup errors compared to dial-based systems.
-
Soft cushion seal improves patient compliance over rigid masks, reducing air leak and oxygen waste.
-
-
Efficiency / cost advantage: Reduces blood gas frequency by 30–40% (per published hospital trials) due to predictable FiO₂; lowers oxygen consumption by up to 25% versus non-rebreather masks at same target saturation.
4. Competitive Advantages
-
Quality consistency: Batch traceability per ISO 13485:2016; each set 100% leak-tested at 15 L/min flow; failure rate <0.2% (industry average ~1.5%).
-
Supply reliability: Dual manufacturing facilities (Asia, Europe) with 99.5% on-time delivery for contracted orders; 90-day safety stock for high-volume buyers.
-
Logistics capability: Palletized, RFID-tagged master cases; direct-to-warehouse or cross-dock options for global B2B buyers.
-
Price competitiveness: Tiered pricing from 10,000 sets/year onward; 12–18% below leading incumbent brands for equivalent spec.
-
Sustainability: PVC recycling program for contracted buyers; packaging uses 80% recycled cardboard; mask components mechanically separable for end-of-life processing.
-
Technical support: Full engineering dossier, IFU in 14 languages, online FiO₂ flow-rate calculator, and 24/7 clinical application helpline.
5. Commercial & Supply Information
-
Minimum order quantity (MOQ): 1,000 complete sets (10 master cases)
-
BULK 20MT loading capacity: 0.72 metric tons per pallet (25 master cases/pallet → 2,500 sets); 20MT = 27 full pallets (approx. 67,500 sets)
-
MT per container:
-
20-ft container: 10–11 MT (approx. 14–15 pallets → 35,000–37,500 sets)
-
40-ft HC container: 20–22 MT (28–30 pallets → 70,000–75,000 sets)
-


Reviews
There are no reviews yet.