Description
1. Product Overview
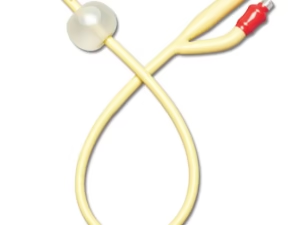
The Nelaton Catheter 12Fr PVC Sterile is a single-use, flexible medical drainage device engineered for short-term bladder catheterization in clinical and surgical settings. Its primary industrial application lies in hospital procurement, home healthcare supply chains, and emergency medical kits where reliable, cost-effective urine drainage is required. The key value proposition is the optimal balance of patient-safe rigidity (12Fr diameter), sterile presentation, and PVC’s proven track record for high-volume, low-cost manufacturing. Strategically, this product is critical for healthcare systems seeking to standardize consumables without compromising aseptic reliability—reducing cross-contamination risks while maintaining procurement efficiency.
2. Key Specifications & Technical Characteristics
-
Material Composition: Medical-grade polyvinyl chloride (PVC), DEHP or non-DEHP options available depending on regulatory requirement; radiopaque line embedded for X-ray visibility
-
Sterility & Grade: EO (ethylene oxide) or gamma sterilized; sterile, single-use, non-pyrogenic; latex-free
-
Physical Characteristics:
-
Color: Transparent with visible radiopaque stripe
-
French size: 12Fr (4.0 mm outer diameter)
-
Length: Standard 40 cm (pediatric) or 45–50 cm (adult) – confirm per batch
-
Tip: Rounded, atraumatic closed tip with 2 lateral eyes
-
Connector: Standard Luer or funnel-type urine drainage connector
-
-
Packaging Options:
-
Individually peel-open sterile pouch (medical paper/PE film)
-
Bulk inner carton: 100 units
-
Master carton: 10–20 inner cartons (1,000–2,000 units per master)
-
-
Shelf Life: 3–5 years from date of sterilization, stored in dry, cool, dark conditions (15–25°C)
3. Core Industrial Applications
-
Primary Industries: Hospital group purchasing organizations (GPOs), surgical centers, long-term care facilities, medical device distributors, emergency response (EMS) suppliers, and home healthcare logistics providers
-
Specific Use Cases:
-
Intermittent or indwelling bladder drainage for post-operative patients
-
Urine output monitoring in ICU or general ward settings
-
Overflow incontinence management in geriatric care
-
Backup catheter for emergency medical kits and disaster relief supplies
-
-
Performance vs. Alternatives:
-
Versus silicone: Lower unit cost, sufficient for short-term use (<14 days); no silicone shedding concerns
-
Versus latex: Zero allergic reaction risk (latex-free)
-
Durability advantage: PVC offers consistent flow rate (lumen integrity) at 12Fr—ideal for non-viscous urine
-
Cost advantage: Up to 60–70% lower than silicone equivalents, enabling high-volume replenishment contracts
-
4. Competitive Advantages
-
Quality Consistency: ISO 13485:2016 certified production; each lot tested for tensile strength, kink resistance, flow rate, and sterilization integrity
-
Supply Reliability: Dual-source raw material (PVC compound) strategy; buffer stock maintained regionally (Asia, EU, NA); lead time 15–25 days for repeat orders
-
Logistics Capability: Export-ready master cartons optimized for palletization; temperature-stable shipment (no cold chain required)
-
Price Competitiveness: Direct factory pricing with tiered volume discounts (2,000+ units), eliminating distributor markup for qualified buyers
-
Sustainability: Non-DEHP PVC options available for EU MDR compliance; reduced packaging waste via high-density master carton design
-
Technical Support: Full sterilization validation documentation (EO residue report, bioburden test); IFU and regulatory filing support for local market registration
5. Commercial & Supply Information
-
Minimum Order Quantity (MOQ):
-
Standard: 10,000 units (10 master cartons)
-
Sample order (non-sterile evaluation): 100 units
-
-
Bulk Loading Capacity (per 20FT container):
-
Approx. 400,000–500,000 individually pouched catheters, depending on master carton dimensions and inner tray design
-
Palletized configuration: 20–22 pallets (EU/Asia standard pallet)
-
-
Note: Sterile units must not be repackaged. Buyer to verify biocompatibility (ISO 10993) for specific patient risk profiles. Volume >1M units/year qualifies for dedicated production line and consignment stock.






Reviews
There are no reviews yet.