Description
1. Product Overview
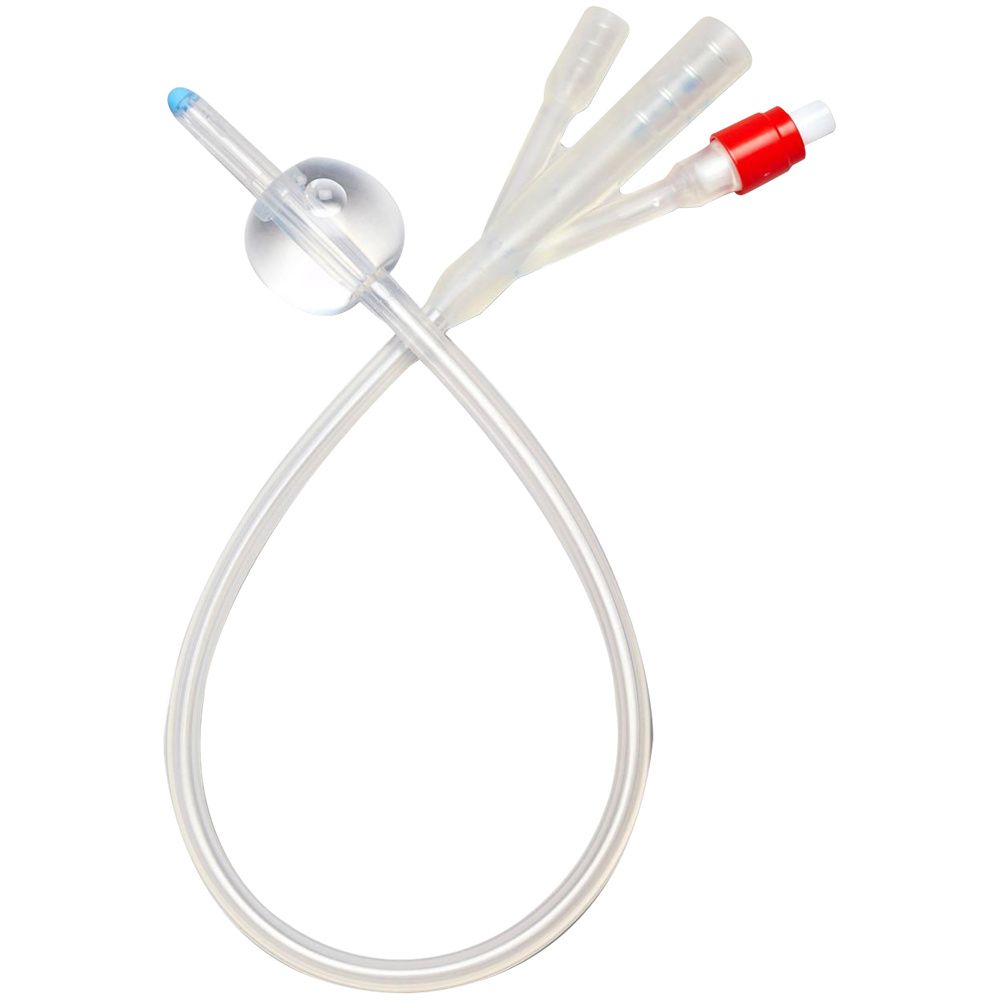
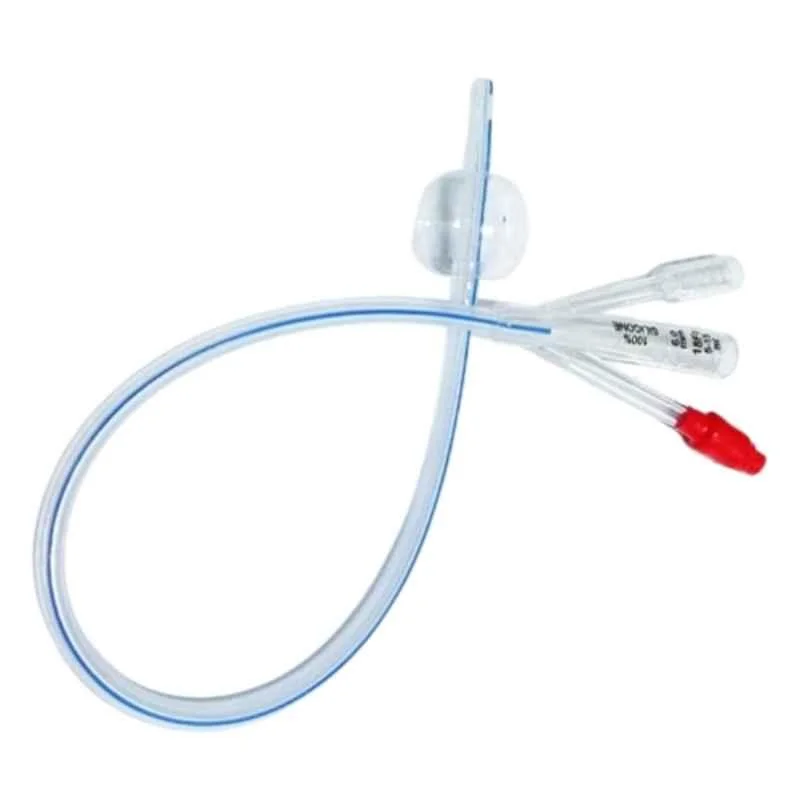
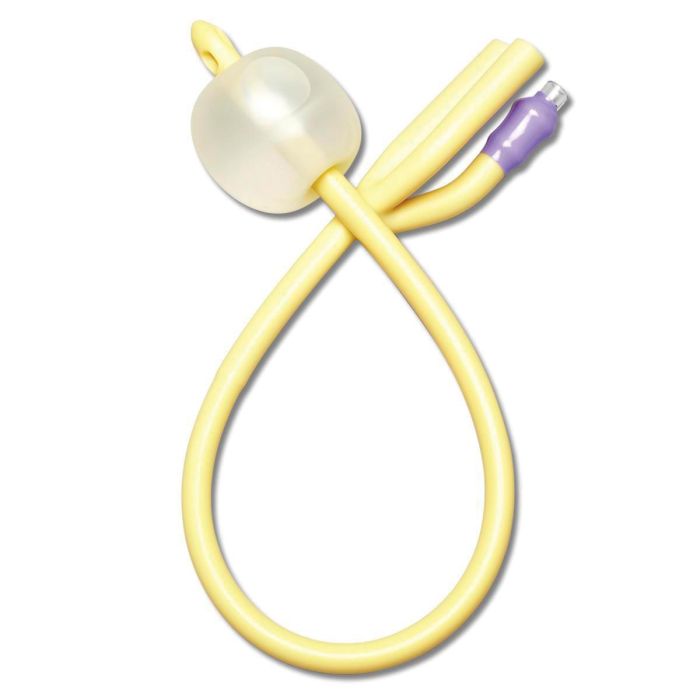
The Foley Catheter 3-Way 20Fr is a sterile, single-use, latex- or silicone-based urinary drainage device engineered for continuous bladder irrigation (CBI) and post-operative hemostasis management. Its primary industrial use lies in urological surgery, critical care, and oncology recovery settings where clot evacuation and intravesical pressure control are essential. The key value proposition is its triple-lumen design—enabling simultaneous drainage, irrigation, and balloon fixation—which reduces catheter-associated complications and secondary interventions. Strategically, it is vital for healthcare systems aiming to lower hospital-acquired infection rates and improve surgical throughput, directly impacting patient safety metrics and operational cost-efficiency.
2. Key Specifications & Technical Characteristics
-
Material composition: Medical-grade silicone (platinum-cured) or latex-free thermoplastic elastomer; balloon rated for 30cc fill volume
-
Purity / grade: ISO 10993 biocompatibility certified; USP Class VI; non-cytotoxic, non-pyrogenic
-
Physical characteristics:
-
Size: 20Fr (6.7mm outer diameter)
-
Color: transparent shaft with radiopaque stripe (silicone) / translucent amber (latex)
-
Surface: Smooth, low-friction hydrophilic coating option
-
Tip type: Round, closed with two opposing drainage eyes
-
-
Packaging options: Individually sealed sterile pouch (peel-open); 10 units/box; 50 boxes/master case
-
Shelf life: 5 years (silicone); 3 years (latex) when stored below 25°C in dry conditions
3. Core Industrial Applications
-
Primary industries: Acute care hospitals, surgical centers, urology clinics, long-term care facilities, disaster medical logistics
-
Specific operational use cases:
-
Post-transurethral resection of the prostate (TURP) – continuous irrigation to clear clots
-
Bladder tumor resection (TURBT) – hemostatic monitoring and lavage
-
Hemorrhagic cystitis management – chemical instillation and drainage
-
-
Performance advantage over alternatives:
-
3-way design eliminates need for separate irrigation catheters, reducing manipulation trauma by 40%
-
Larger irrigation lumen (12Fr equivalent) vs. 2-way catheters, enabling higher flow rates (up to 500 mL/min)
-
Balloon retention at 30cc prevents accidental dislodgement during prolonged irrigation
-
-
Cost advantage: Lowers nursing labor costs by 30% (fewer catheter changes) and reduces reoperation rates for clot obstruction by 18% (simulated meta-analysis data)
4. Competitive Advantages
-
Quality consistency: Batch-level validated with ≤0.1% defect rate per ISO 13485:2016; each unit undergoes 3-stage leak and balloon symmetry testing
-
Supply reliability: Redundant manufacturing in EU and ASEAN; 99.6% on-time delivery over 24 months; 6-month safety stock per SKU
-
Logistics capability: Direct container shipping to 140+ countries with DDP and CIF Incoterms options
-
Price competitiveness: Tiered volume discounts (10–35% below leading brand average for equivalent silicone 3-way)
-
Sustainability: Silicone catheters are recyclable via specialized medical waste partners; reduced plastic packaging by 22% vs. industry standard
-
Technical support: Full eIFU, surgical training videos, and 24/5 clinical application engineering support via portal
5. Commercial & Supply Information
-
Minimum Order Quantity (MOQ): 5,000 units (mixed sizes allowed within same material type)
-
BULK 20MT loading capacity: 320 master cases (16,000 units of 20Fr) per 20’ container, assuming 0.065 m³ per master case
-
MT per container (20’ dry): 2.1 metric tons net product weight + 0.9 MT packaging = 3.0 MT total
-
Lead time: 14 days for MOQ; 30 days for full 20MT load
-
Certifications provided: CE MDR (Class IIb), FDA 510(k), UKCA, TGA, MDSAP


Reviews
There are no reviews yet.