Description
1. Product Overview
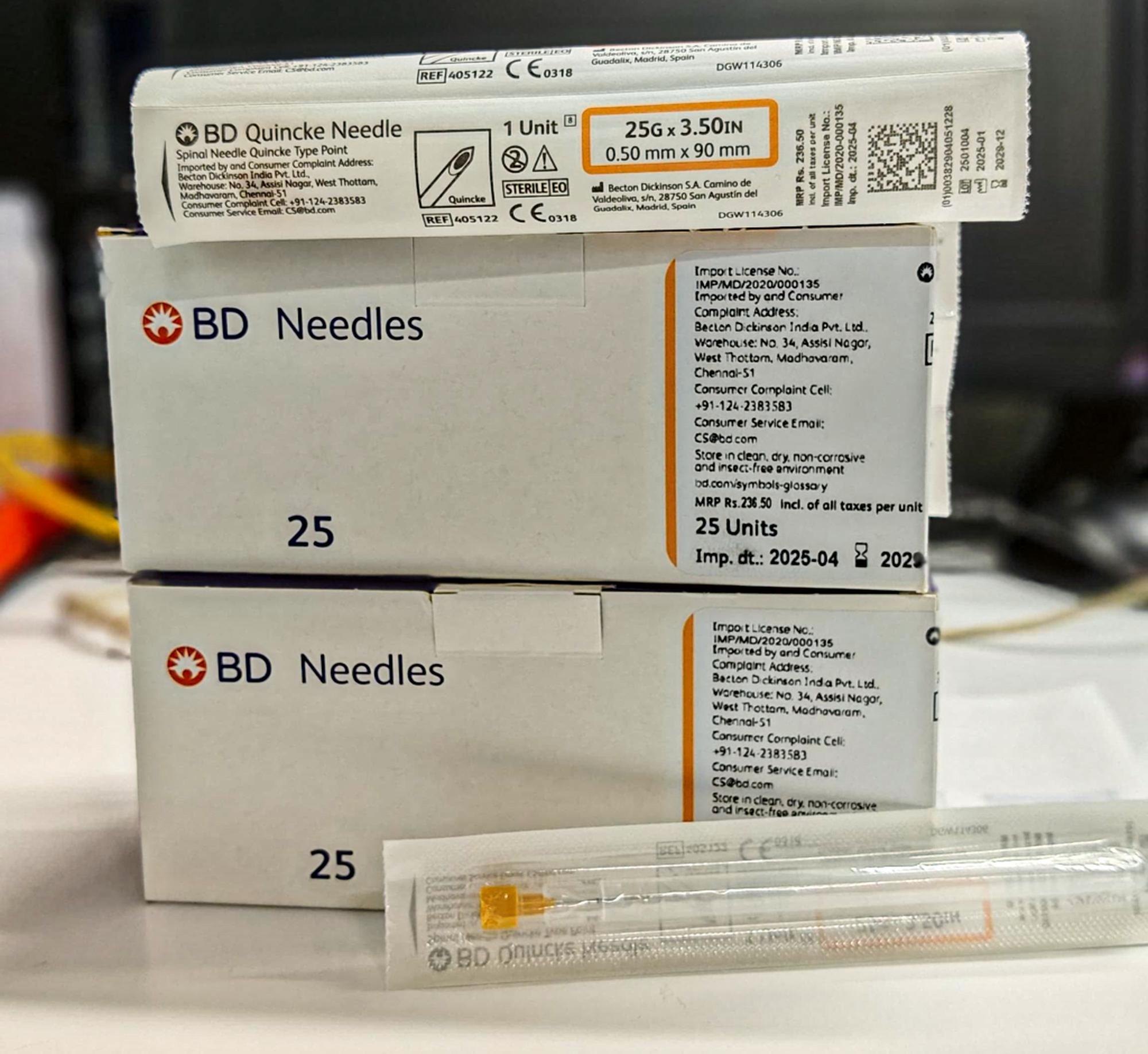
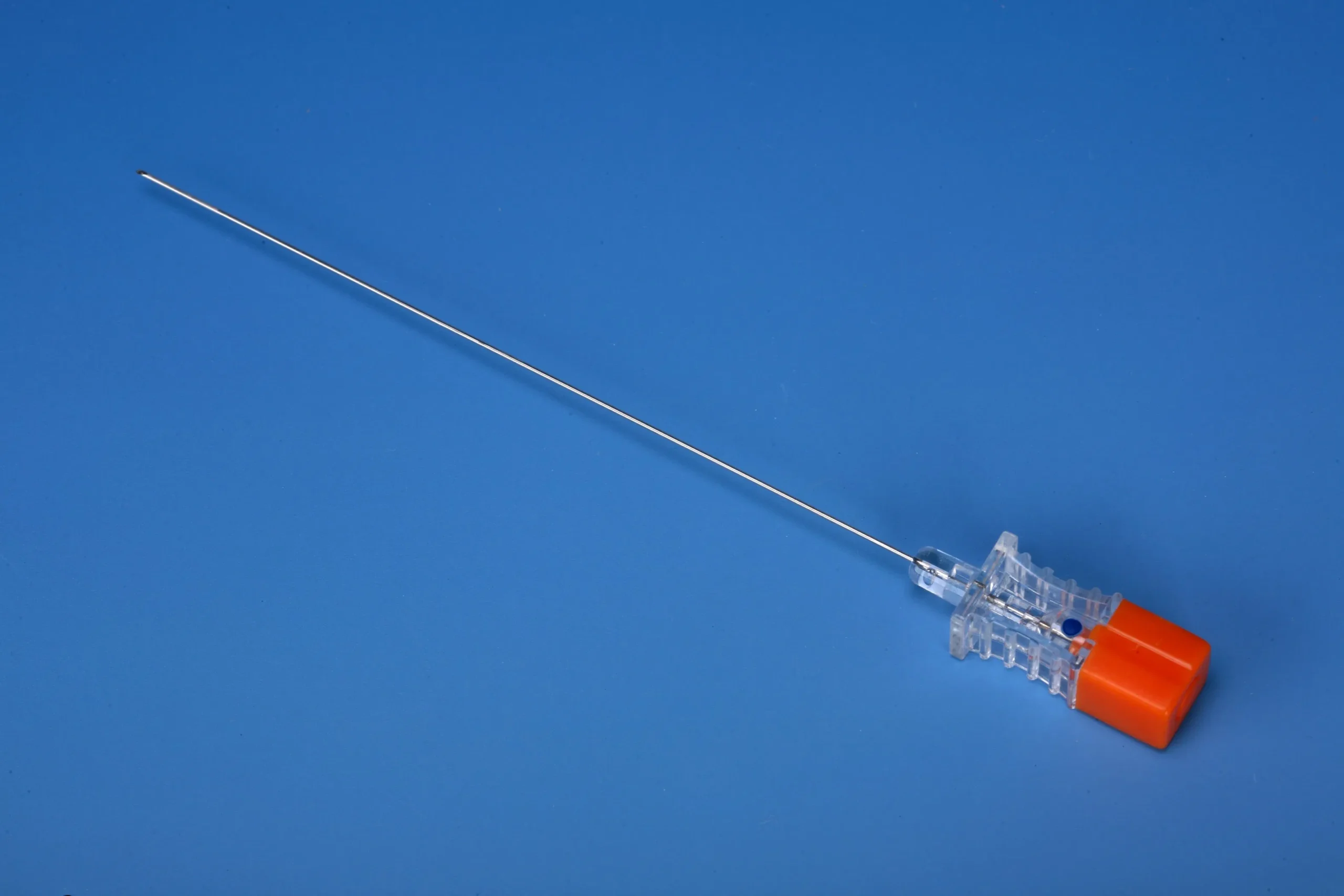
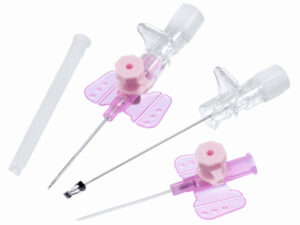
The Spinal Needle Quincke Type 25G is a precision-engineered, single-use medical device designed for subarachnoid block administration in diagnostic and therapeutic spinal procedures. Its primary industrial application lies within hospital anesthesia departments, ambulatory surgical centers, and pain management clinics requiring reliable cerebrospinal fluid (CSF) access. The key value proposition is an optimal balance between minimal dural trauma (due to 25G gauge) and consistent CSF flow rate, reducing post-dural puncture headache (PDPH) incidence. Strategically, this product addresses the growing global demand for safer, lower-morbidity spinal anesthesia devices, positioning it as a critical supply chain component for healthcare systems prioritizing patient outcomes and procedural efficiency.
2. Key Specifications & Technical Characteristics
-
Material composition: Medical-grade stainless steel hub and cannula; hub bonded with USP Class VI medical adhesive; stylet made from hardened stainless steel
-
Needle gauge: 25G (ISO 9626:2016)
-
Point geometry: Quincke (tri-bevel cutting point) with stylus matched to cannula lumen for minimal tissue coring
-
Hub connection: Luer lock (ISO 594-1 compliant), color-coded in orange (25G standard)
-
Shaft markings: Depth graduation bands at 5 mm intervals from 30 mm to 90 mm
-
Sterility: Ethylene oxide (EtO) sterilized; SAL 10⁻⁶
-
Packaging options:
-
Individually sealed Tyvek/polyethylene peel pouch
-
50 units / inner box
-
500 units / master carton
-
-
Shelf life: 5 years from date of manufacture (unopened, controlled storage: 10°C–30°C, <80% RH)
-
Single-use: Yes, non-pyrogenic, latex-free, DEHP-free
3. Core Industrial Applications
-
Primary industries: Hospital anesthesia, interventional radiology, neurology (CSF sampling), veterinary anesthesia (large animal spinal procedures)
-
Specific operational use cases:
-
Elective orthopedic and urologic surgeries requiring spinal anesthesia
-
Diagnostic lumbar puncture for CSF analysis (e.g., suspected meningitis, subarachnoid hemorrhage)
-
Intrathecal drug delivery (chemotherapy, opioids, contrast media)
-
-
Performance advantage over alternatives:
-
Compared to 22G–24G: 25G significantly reduces PDPH rate (studies show ~5-10% vs. 20-40% for larger gauges) while maintaining CSF return within 5–15 seconds.
-
Compared to pencil-point (e.g., Whitacre, Sprotte): Quincke tip provides cleaner entry through ligamentum flavum with lower insertion force and less tactile uncertainty for anesthesia providers.
-
-
Cost/efficiency advantage: Lower per-unit cost than specialized atraumatic tips, compatible with standard introducer needles (22G x 1.5″), and reduces post-procedure nursing time and unplanned hospital readmissions for PDPH treatment.
4. Competitive Advantages
-
Quality consistency: Each batch subjected to 3-point inspection – tip integrity (200x magnification), stylet fit (pull force ≤0.5N), and flow rate (≥0.5 mL/min saline at 50 cm H₂O).
-
Supply reliability: Dual manufacturing sites (ISO 13485:2016, MDSAP certified); safety stock of 2M+ units maintained in regional hubs (NA, EU, APAC).
-
Logistics capability: 72-hour dispatch for stock orders; temperature-controlled shipping for global destinations; real-time tracking via supplier portal.
-
Price competitiveness: Tiered pricing reduces per-unit cost by up to 22% at annual volumes of 100,000+ units, without compromising on bevel polish or hub molding tolerances.
-
Technical support: Full IFU in 14 languages; lot traceability to raw material certificates; access to clinical case studies comparing PDPH rates across gauges.
-
Sustainability: 100% recyclable sterile barrier packaging; manufacturing powered with 35% renewable energy; zero latex/DEHP eliminates disposal restrictions in EU/NA.
5. Commercial & Supply Information
-
Minimum order quantity (MOQ):
-
Initial order: 2,000 units (4 master cartons)
-
Repeat order: 1,000 units
-
-
Bulk 20MT loading capacity: Not applicable (product classified as medical device – high volume, low weight).
Equivalent density: 500 master cartons (250,000 needles) = approx. 1.2 MT (volume ~ 12 CBM). -
Loading capacity per 20FT container: Up to 1,800 master cartons (900,000 needles) when palletized with mixed SKUs or 2,200 master cartons (1.1M needles) in master carton direct stacking.
-
Lead time:
-
Stock ≤50,000 units: 5 working days
-
≥50,000 units (manufacturing run): 21 working days
-
-
Incoterms offered: EXW, FOB (Shanghai / Rotterdam / Houston), CIF (major global ports)


Reviews
There are no reviews yet.