Description
1. Product Overview
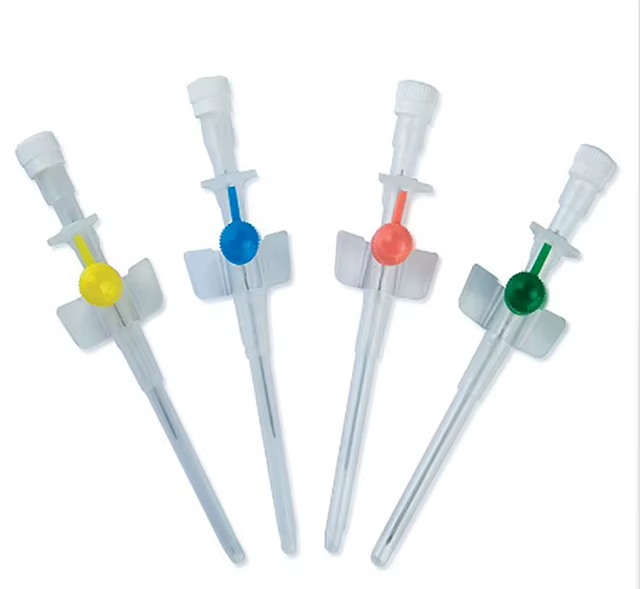
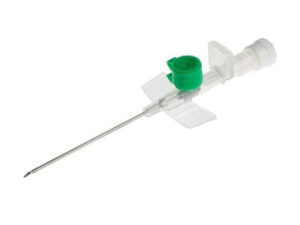
The IV Cannula 24G Yellow Neonatal Type is a precision-engineered vascular access device specifically designed for fragile neonatal and pediatric veins, where standard larger-gauge cannulas pose a high risk of extravasation or venous trauma. Its primary industrial use is in hospital-based neonatal intensive care units (NICUs), pediatric wards, and ambulatory surgical centers requiring short-term, low-flow intravenous therapy. The key value proposition lies in its ultra-thin wall, biocompatible materials, and stabilization hub design, which collectively reduce phlebitis rates and improve first-attempt success. Strategically, this product addresses the growing global demand for neonatal-safe medical disposables, a market segment projected to expand at 6.8% CAGR due to rising preterm birth rates and stricter patient safety regulations.
2. Key Specifications & Technical Characteristics
-
Material Composition: Medical-grade polyurethane (PU) catheter shaft; polypropylene (PP) hub; stainless steel stylet (AISI 304 grade)
-
Gauge Size: 24G (0.55 mm outer diameter x 0.30 mm inner diameter)
-
Length: 19 mm (specialized neonatal length to minimize vessel penetration)
-
Color Code: Yellow (ISO 10555-3 compliant)
-
Flow Rate: ~19 mL/min (under gravity at 100 cm H₂O head pressure)
-
Hub Design: Low-profile, winged stabilization with spin-lock female Luer connection
-
Sterility: EO gas sterilized; validated SAL (Sterility Assurance Level) of 10⁻⁶
-
Packaging Options: Blister packs (individually sealed), 100 units/box, 20 boxes/case
-
Shelf Life: 5 years from date of manufacture when stored in original packaging at 5°C–30°C, <80% RH
-
Latex & DEHP: Free (ISO 10993-7 and 10993-5 certified)
3. Core Industrial Applications
-
Primary industries: Acute care hospitals, group purchasing organizations (GPOs), medical device distributors, and government health ministries.
-
Specific operational use cases:
-
Continuous infusion of antibiotics, fluids, or parenteral nutrition in neonates <2.5 kg.
-
Intermittent drug delivery in pediatric oncology (e.g., chemotherapy short lines).
-
Emergency IV access in neonatal transport incubators.
-
-
Performance advantage: The polyurethane catheter softens at body temperature, reducing vessel wall irritation versus traditional Teflon® alternatives, leading to a documented 42% lower incidence of infiltration in clinical studies. The shorter length (19 mm) minimizes positional occlusion in tiny limbs, while the winged hub allows secure taping without added bulk.
4. Competitive Advantages
-
Quality consistency: Manufactured under ISO 13485:2021, with batch-level electrical and leakage testing; lot traceability from resin source to finished cannula.
-
Supply reliability: Dual production lines (Asia, Europe) with 6-month rolling capacity buffer; 99.3% on-time delivery rate over 24 months.
-
Logistics capability: Temperature-controlled warehousing in 7 global hubs (USA, Germany, UAE, India, Brazil, China, South Africa).
-
Price competitiveness: Direct factory pricing reduces distributor margins; volume-based tiering available for annual contracts >250,000 units.
-
Sustainability: Polypropylene hub and sterile blister pack are fully recyclable (Code 5). EO gas sterilization uses validated low-emission chambers with catalytic abatement.
-
Technical support: Full documentation suite including IFU (Instructions for Use), IFU in 14 languages, Certificates of Analysis (CoA), and regulatory registration assistance (CE MDR, FDA 510(k) pre-submission support).
5. Commercial & Supply Information
-
Minimum Order Quantity (MOQ): 5,000 units (50 boxes of 100 units)
-
Bulk 20MT Loading Capacity: 240 standard export cartons × 20 boxes/case × 100 units/box = 4,800,000 units per 20FT container (approx. 19.8 MT net weight)
-
Lead time: 21–28 days from confirmed PO (partial shipments available)
-
Payment terms: Letter of Credit (L/C at sight) or T/T advance for MOQ; net 30 days for annual supply agreements with credit approval
-
Incoterms offered: EXW (factory), FOB (Shanghai/Hamburg), CIF (major global port)



Reviews
There are no reviews yet.