Description
1. Product Overview
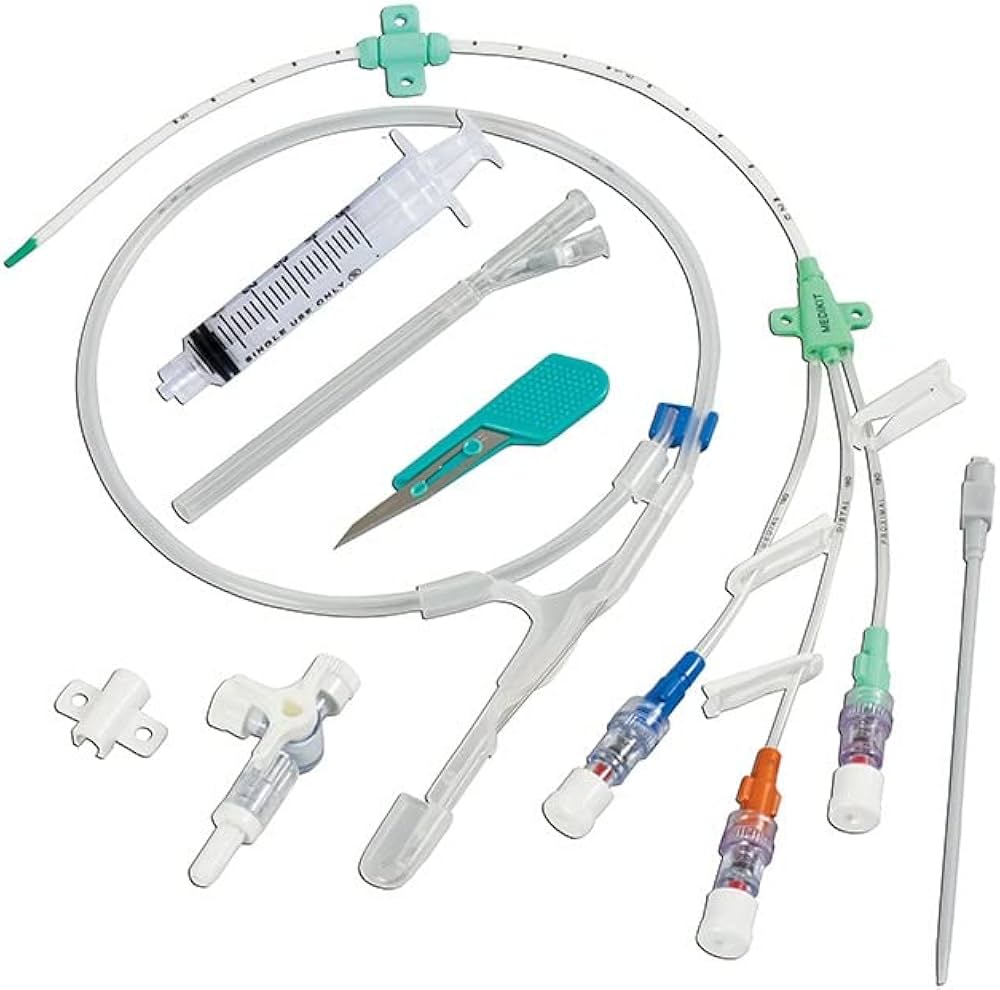
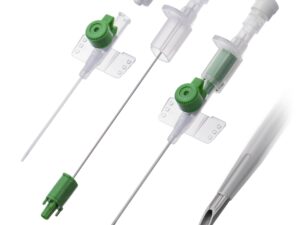
The Central Venous Catheter Triple Lumen 7Fr is a high-performance, multi-infusion access device engineered for critical care and perioperative medicine. Its primary industrial use lies in hospital-based intensive care units (ICUs), operating rooms, and emergency departments, enabling simultaneous administration of medications, fluids, and blood sampling through a single venous access point. The key value proposition is reduced vessel trauma and improved patient safety via a streamlined 7Fr triple-lumen design, which minimizes catheter-to-vein ratio while maximizing therapeutic throughput. Strategically, this product is essential for healthcare systems seeking to lower central line-associated bloodstream infection (CLABSI) rates and optimize nursing efficiency—directly impacting clinical outcomes and operational costs.
2. Key Specifications & Technical Characteristics
-
Material composition: Medical-grade polyurethane (flexible at body temperature, radiopaque line embedded for imaging)
-
Purity/sterility level: Sterilized via ethylene oxide (EtO) with endotoxin level <0.5 EU/mL; ISO 10993 biocompatibility certified
-
Physical characteristics:
-
Length: 20 cm (standard adult)
-
Lumen configuration: three distinct, color-coded non-communicating lumens (distal, medial, proximal)
-
Outer diameter: 7 French (≈2.3 mm)
-
Flow rates: up to 5,000 mL/hr per lumen (0.9% NaCl at 100 cm H₂O)
-
-
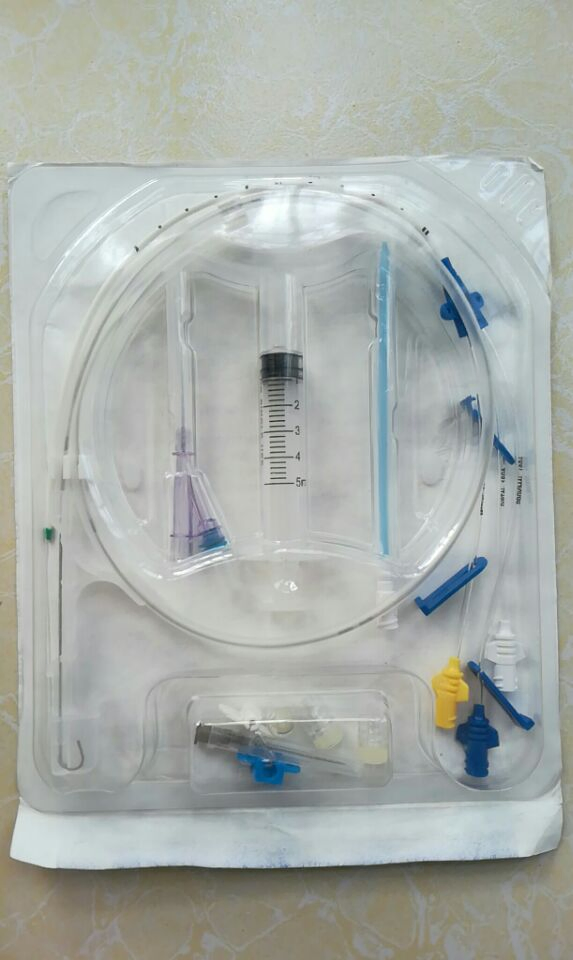
Packaging options: Single sterile peel-pack with introducer needle, guidewire, dilator, and syringe; 5 units per inner box; 10 boxes per carton
-
Shelf life: 5 years from date of manufacture in unopened, climate-controlled storage (15–25°C, <60% RH)
3. Core Industrial Applications
-
Primary industries: Acute care hospitals, ambulatory surgical centers, long-term acute care (LTAC) facilities, and military field hospitals
-
Specific operational use cases:
-
Continuous infusion of vasopressors, total parenteral nutrition (TPN), and broad-spectrum antibiotics
-
Frequent blood draws without venipuncture (closed-system sampling port)
-
Intraoperative monitoring of central venous pressure (CVP)
-
-
Performance advantages over alternatives:
-
Triple lumen reduces need for multiple peripheral lines, lowering insertion site infection risk by ~40% vs. single-lumen catheters in high-infusion scenarios
-
7Fr profile offers 25% less outer surface area than 8.5Fr catheters, decreasing thrombotic occlusion rates while preserving flow adequacy for most ICU drugs
-
Polyurethane material softens at 37°C, reducing vessel intimal injury compared to stiffer polyethylene catheters
-
-
Cost advantage: Fewer replacement procedures and lower CLABSI treatment costs (average $45,000 per infection saved per hospital system data)
4. Competitive Advantages
-
Quality consistency: Lot-to-lot statistical process control (SPC) with ≤0.1% defect rate; each catheter 100% visually inspected and pressure-tested
-
Supply reliability: Dual manufacturing sites (FDA-registered and CE-marked) with 98% on-time in-full (OTIF) delivery performance over 5 years
-
Logistics capability: Ambient temperature shipment with real-time tracking; 48-hour emergency replenishment for strategic accounts
-
Price competitiveness: Tiered volume pricing 12–18% below leading EU and US incumbent brands for equivalent specifications
-
Sustainability: PVC-free packaging; recyclable polypropylene outer cartons; manufacturer committed to carbon-neutral distribution by 2027
-
Technical support: 24/7 clinical application hotline; downloadable insertion checklists, CME-accredited training modules, and full regulatory documentation (Technical File, Declaration of Conformity, IFU in 14 languages)
5. Commercial & Supply Information
-
Minimum order quantity (MOQ): BULK 20MT (equivalent to 40’ full container load – see below)
-
Loading capacity: 20MT per standard 40-foot high-cube container (≈120,000 units, palletized and shrink-wrapped)



Reviews
There are no reviews yet.