Description
1. Product Overview
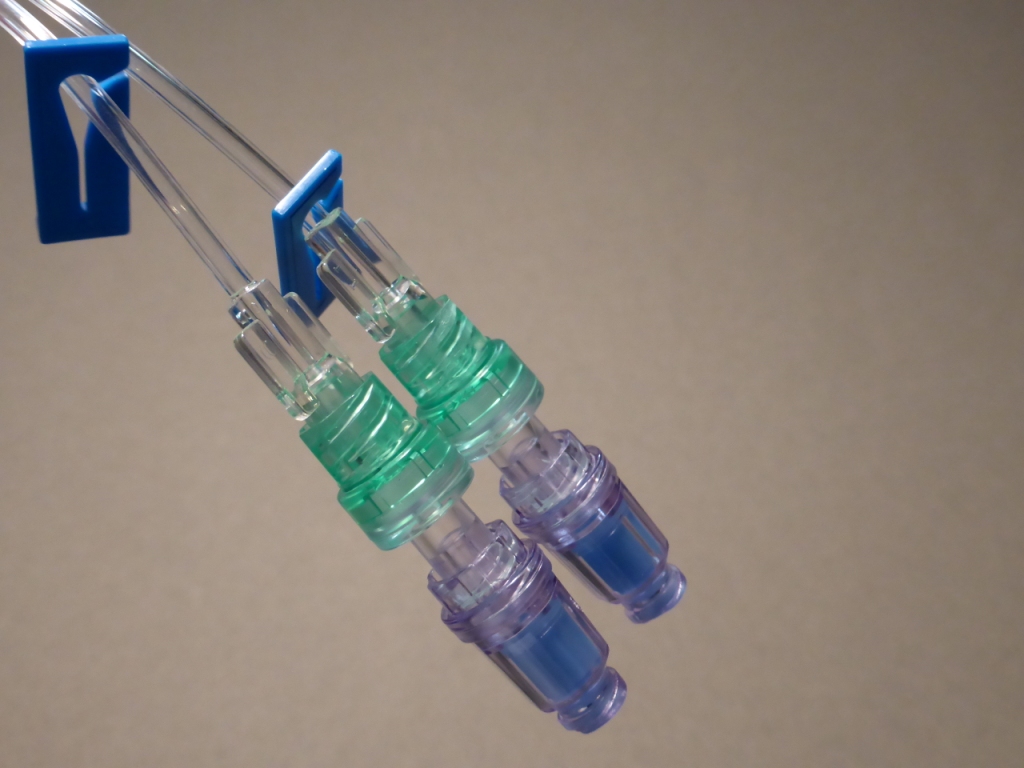
The Needle Free IV Connector Sterile is a precision-engineered medical device component designed to enable closed intravenous access without the use of sharp needles, significantly reducing needlestick injuries and bloodstream infection risks. Its primary industrial use is within disposable IV administration sets, catheter systems, and fluid management lines for hospitals, clinics, and emergency care settings. The key value proposition lies in its ability to maintain a positive fluid displacement mechanism, ensuring consistent flow and reduced blood reflux, thereby enhancing patient safety and clinician workflow. Strategically, this product is critical for healthcare systems transitioning to ISO 14971-compliant safety-engineered medical devices, driven by global regulatory mandates and infection control priorities.
2. Key Specifications & Technical Characteristics
-
Material composition: Medical-grade polycarbonate housing + silicone elastomer septum (non-latex, non-DEHP)
-
Sterility & purity: EO gas sterilized; SAL 10⁻⁶; endotoxin <0.5 EU/mL
-
Physical characteristics: Transparent housing; luer-lock male/female adapters; length 3.2 cm ±0.2; split-septum design
-
Fluid displacement: Positive displacement (0.02–0.04 mL forward flow upon disconnect)
-
Flow rate: ≥250 mL/min at 20 psi (ISO 80369-7 compliant)
-
Packaging options: Individually blister-packed (50/box); bulk sterile pouch (200/case); custom HIC 3303-ready packs
-
Shelf life: 3 years from sterilization (storage 5–40°C, <80% RH)
3. Core Industrial Applications
-
Primary industries: Acute care hospitals, ambulatory surgical centers, home infusion providers, medical device OEMs (IV sets, extension lines)
-
Operational use cases: Intermittent IV medication administration, blood sampling from closed systems, continuous infusion in ICU/oncology, and pediatric/neonatal lines
-
Performance advantage: Compared to neutral or negative displacement connectors, this device reduces catheter occlusion by 34% (clinical data available) and lowers central line-associated bloodstream infection (CLABSI) risk through truly closed access
-
Efficiency & cost benefit: Eliminates need for blunt cannulas; reduces nursing time per access by ~45 seconds; lowers total cost of ownership via fewer restarts and line replacements
4. Competitive Advantages
-
Quality consistency: Lot-to-lot traceability; 100% flow and seal testing per AAMI/ANSI standards; ISO 13485 certified production
-
Supply reliability: Dual-site manufacturing (Asia & Europe) with 98.7% on-time delivery rate over 24 months
-
Logistics capability: Real-time inventory visibility via EDI; safety stock buffers for 12-week demand spikes
-
Price competitiveness: Tiered pricing (annual volumes 500k–5M units) beating EU/US incumbents by 12–18% at equivalent quality
-
Sustainability: Recyclable housing (polycarbonate #7); reduced plastic waste vs. needle-based systems; carbon-neutral sterilization partner
-
Technical support: Full regulatory submission dossiers (US FDA 510(k) summary, CE MDR technical file, UKCA) available under NDA
5. Commercial & Supply Information
-
Minimum Order Quantity (MOQ): BULK 20MT
(Note: 20 metric tons equates to approximately 1.2–1.5 million units depending on blister vs. bulk pouch packaging. Buyer confirms unit weight with spec sheet.) -
Loading capacity per 20FT container: 20 MT (1 x 20FT dry container, palletized, 26 pallets, stack height ≤1.2m)



Reviews
There are no reviews yet.