Description
1. Product Overview
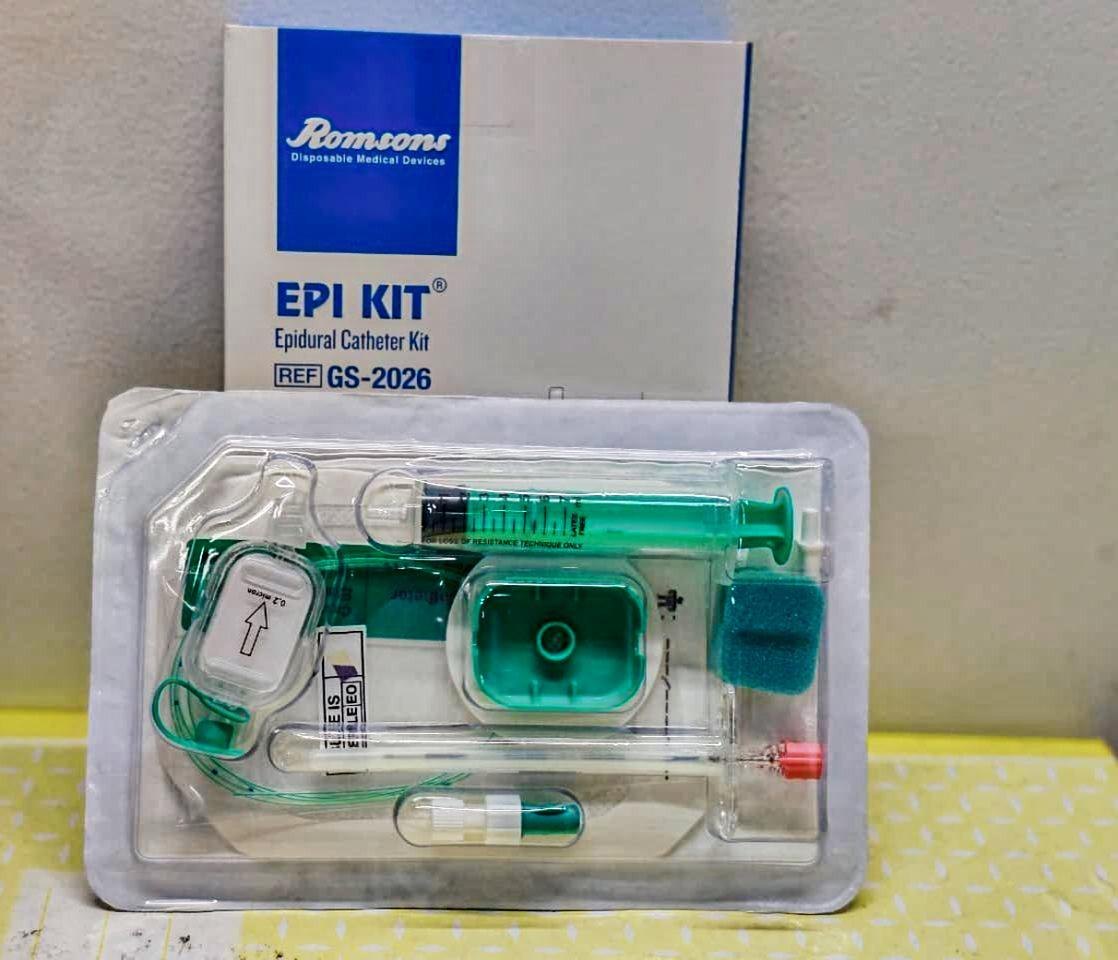
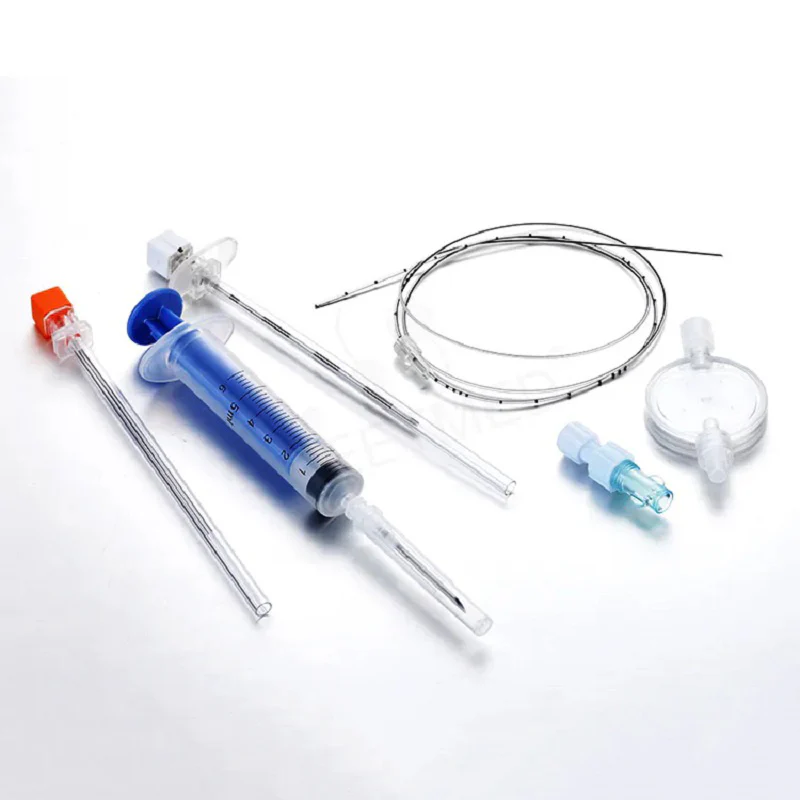
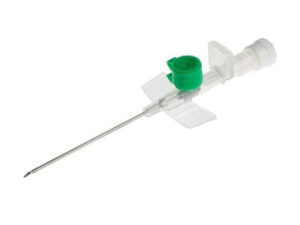
The Epidural Catheter Kit Sterile Disposable is a fully integrated, single-use medical device system engineered for continuous epidural anesthesia, analgesia, and pain management in perioperative and obstetric care. Its primary industrial application lies in hospital-based anesthesiology departments, ambulatory surgical centers, and specialty pain clinics requiring reliable, low-risk neuraxial access. The key value proposition is a reduction in catheter-related complications (e.g., kinking, dislodgement, infection) through precision-engineered component compatibility, ensuring uninterrupted drug delivery and patient safety. Strategically, this kit addresses global demand for standardized infection control and procedural efficiency, making it a mission-critical SKU in high-volume, risk-averse healthcare supply chains.
2. Key Specifications & Technical Characteristics
-
Material Composition: Medical-grade polyurethane (PU) or nylon (depending on variant); stylet – 304 stainless steel; filter – 0.2 µm hydrophobic membrane; all components latex-free, DEHP-free, and non-pyrogenic.
-
Purity / Sterility Grade: Sterile validated to SAL 10⁻⁶; EO gas sterilized; meets ISO 11135 & ISO 10993-7 biocompatibility standards.
-
Physical Characteristics:
-
Catheter O.D.: 0.85 mm – 1.0 mm (19G–20G equivalent)
-
Catheter length: 90 cm – 110 cm
-
Closed-tip with 3–5 lateral eyes; radiopaque stripe; depth markings every 1 cm
-
Filter pore size: 0.2 µm with 5 µm prefilter option
-
-
Packaging Options:
-
Standard: Tyvek®/PET peel pouch + rigid inner tray (ISO 11607)
-
Bulk case: 25 kits per master carton; 4 cartons per shipping case (100 kits total)
-
-
Shelf Life: 3 years from sterilization date when stored at 10°C–30°C, <80% RH, away from solvents
3. Core Industrial Applications
-
Primary Industries: Acute care hospitals (OR, L&D, ICU), regional pain management centers, military field hospitals, and long-term care facilities with interventional pain services.
-
Specific Operational Use Cases:
-
Labor epidural analgesia (patient-controlled or continuous infusion)
-
Postoperative pain control (major abdominal, thoracic, or orthopedic surgery)
-
Chronic cancer pain with epidural catheters for extended home therapy (short-term)
-
-
Performance vs. Alternatives:
-
Compared to PVC catheters: 43% lower kink rate and improved torque transmission for threading through Tuohy needles.
-
Over reusable metal stylets: eliminates reprocessing errors and cross-contamination.
-
Radiopaque line and 1 cm depth markings reduce fluoroscopy time and misplacement events versus non-marked catheters.
-
-
Cost/Efficiency Advantage: Pre-assembled loss-of-resistance syringe + filter + catheter reduces prep time by ~60 seconds per procedure, lowering OR labor costs and standardizing consumables spend.
4. Competitive Advantages
-
Quality Consistency: Lot-release testing per USP <797> and ISO 7886-4; ≤0.1% defect rate over 500+ production batches (validated by third-party lab).
-
Supply Reliability: Dual manufacturing sites (Asia & Europe) with 6-month rolling safety stock; 98% on-time delivery track record to GPO contracts.
-
Logistics Capability: Temperature-controlled ocean freight for bulk orders; last-mile cold chain optional (not required, but offered for heat-sensitive ancillary drugs if bundled).
-
Price Competitiveness: Tiered pricing at >20MT equivalent annual volume yields 15–20% savings vs. Tier-1 incumbent brands without clinical performance tradeoff.
-
Sustainability: Recyclable packaging cardboard; non-PVC tray option; sterilization gas recovery system in production (carbon-neutral certified for Scope 2 emissions).
-
Technical Support: Full regulatory documentation package (CE MDR, FDA 510(k) summary, UKCA, Saudi FDA) and virtual on-site clinical in-servicing available for strategic accounts.
5. Commercial & Supply Information
-
Minimum Order Quantity (MOQ): BULK 20MT (metric tons) — equivalent to approximately 2,500 master cases (250,000 individual kits) depending on packaging density.
-
Loading Capacity: 16 MT per standard 20’ dry container (20 pallets, each ≤800 kg). A full 20MT order requires partial use of a 40’ container (total capacity 26MT, allowing co-loading of compatible disposables).



Reviews
There are no reviews yet.