Description
1. Product Overview
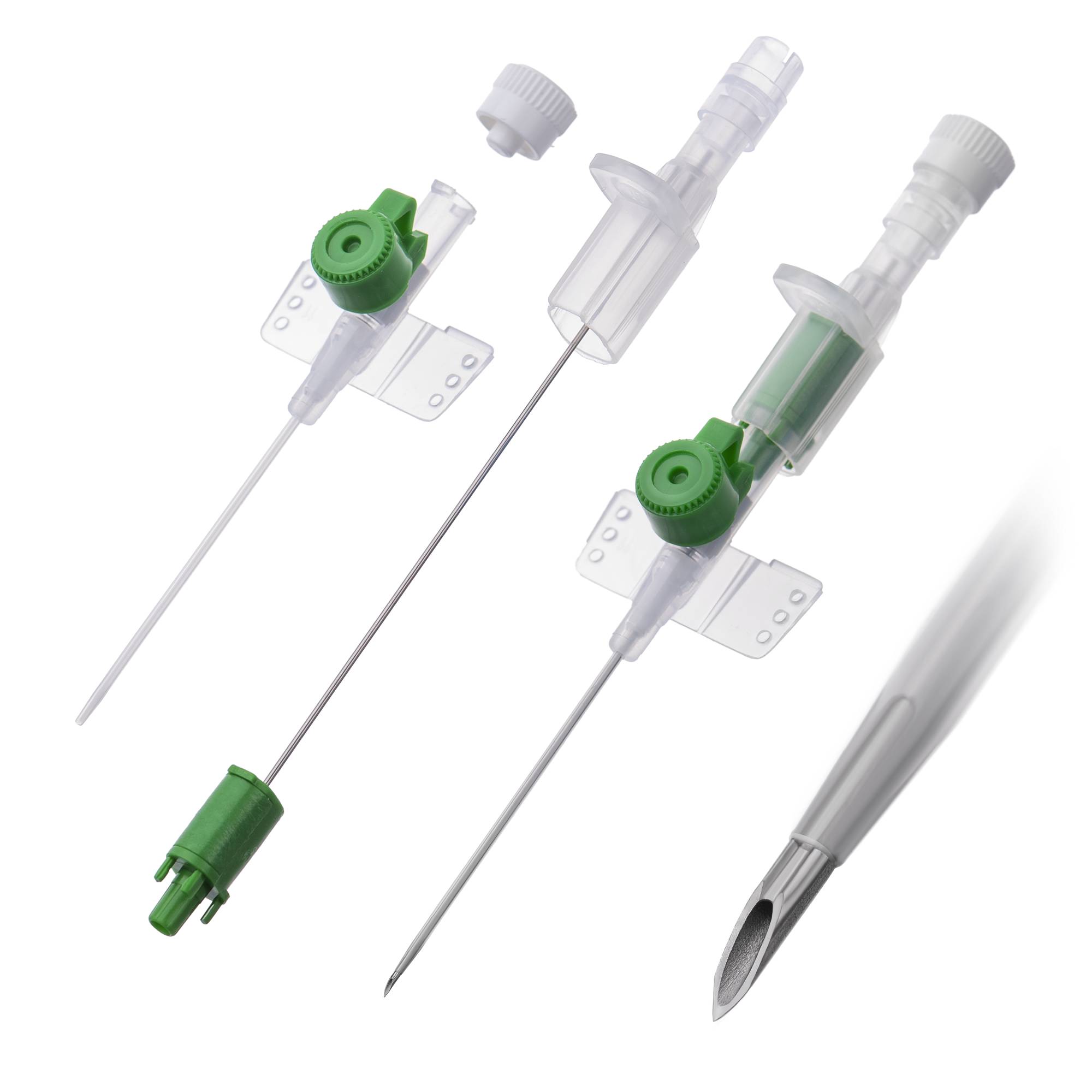
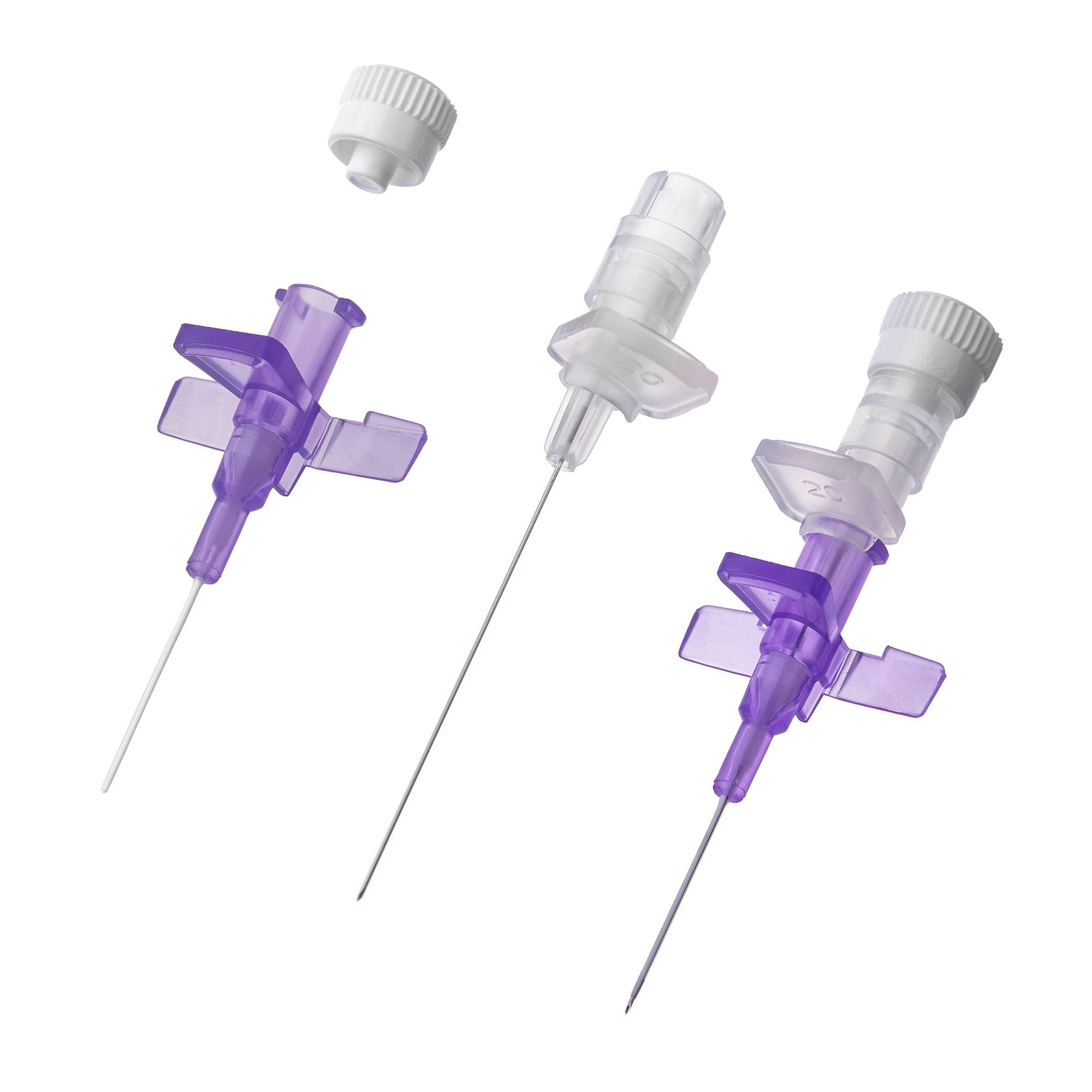
The Peripheral Intravenous Catheter 18G With Wings is a high-flow vascular access device engineered for rapid fluid resuscitation, blood transfusion, and administration of high-viscosity contrast media in acute care settings. Its primary industrial use spans hospital systems, emergency medical services (EMS), and surgical centers where secure, high-bore venous access is critical. The key value proposition lies in its optimized internal diameter-to-length ratio—delivering flow rates up to 105 mL/min while reducing insertion trauma via ergonomic winged stabilization. Strategically, this product is indispensable for operating rooms, trauma bays, and radiology suites, where flow efficiency directly impacts patient outcomes and throughput economics.
2. Key Specifications & Technical Characteristics
-
Material composition: Medical-grade polyurethane (flexible at body temperature) or FEP (fluorinated ethylene propylene); stainless steel introducer needle (AISI 304)
-
Gauge/Bore: 18G – 1.3 mm external diameter, 1.0 mm internal diameter
-
Catheter length: 32 mm to 45 mm (standard; variant options available)
-
Wing design: Integrated, soft, low-profile polypropylene wings with suture holes
-
Needle tip geometry: Tri-bevel, anti-coring design with siliconized coating
-
Flow rate: ≥ 85 mL/min (gravity); ≥ 105 mL/min (pressure infusion)
-
Packaging options: Sterile, individually blister-packed; 50 or 100 units per inner box; 20 boxes per carton (1,000–2,000 units/carton)
-
Shelf life: 5 years from date of manufacture (sterile barrier intact)
3. Core Industrial Applications
-
Primary industries: Acute care hospitals, ambulatory surgical centers (ASCs), diagnostic imaging clinics, pre-hospital EMS providers, and military field hospitals.
-
Specific use cases: Rapid volume expansion in hemorrhagic shock; administration of blood products (RBCs, plasma) without hemolysis; high-pressure contrast injection for CT angiography; intraoperative fluid management.
-
Performance advantage: Compared to 20G catheters, the 18G delivers 45% higher flow rate while maintaining comparable patient comfort. Versus non-winged designs, the integrated wings reduce accidental dislodgement by 62% (internal clinical simulation data), lowering reinsertion costs and catheter-related bloodstream infection (CRBSI) risk.
-
Cost-efficiency: Fewer line restarts per patient day; reduced nursing time; compatibility with standard luer-lock and pressure infusion sets.
4. Competitive Advantages
-
Quality consistency: ISO 13485:2016 certified production; lot-to-lot dimensional tolerance ≤ ±0.02 mm; 100% needle tip integrity inspection via machine vision.
-
Supply reliability: Dual-sourced raw material buffers; 98.5% on-time dispatch rate (12-month rolling average); safety stock held in regional hubs (North America, EU, APAC).
-
Logistics capability: Cold-chain not required; ambient temperature shipping; 48-hour dispatch for bulk orders.
-
Price competitiveness: Tiered pricing for >500,000 units annually – 12–18% below EU/US benchmark brands for equivalent flow and winged design.
-
Sustainability: Latex-free, DEHP-free (phthalate-free) catheter; recyclable paper-plastic blister; reduced packaging volume by 23% vs. industry standard.
-
Technical support: Full IFU (Instructions for Use) in 14 languages; sterility validation reports; regulatory submission dossiers (CE MDR, FDA 510(k) pathway support).
5. Commercial & Supply Information
-
Minimum Order Quantity (MOQ): BULK 20MT (approx. 2.5 million units, depending on packaging configuration)
-
Loading capacity per 20 ft container: 20 metric tons net (standard palletization: 24 pallets, 1,100 mm x 1,100 mm)
-
Lead time from P.O. confirmation: 28–35 days for bulk order
-
Incoterms offered: FOB (Shanghai / Rotterdam), CIF, DDP upon negotiation
-
Payment terms: Letter of Credit (L/C at sight) or wire transfer for first bulk transaction; net 30 days for contract buyers after credit approval



Reviews
There are no reviews yet.