Description
1. Product Overview
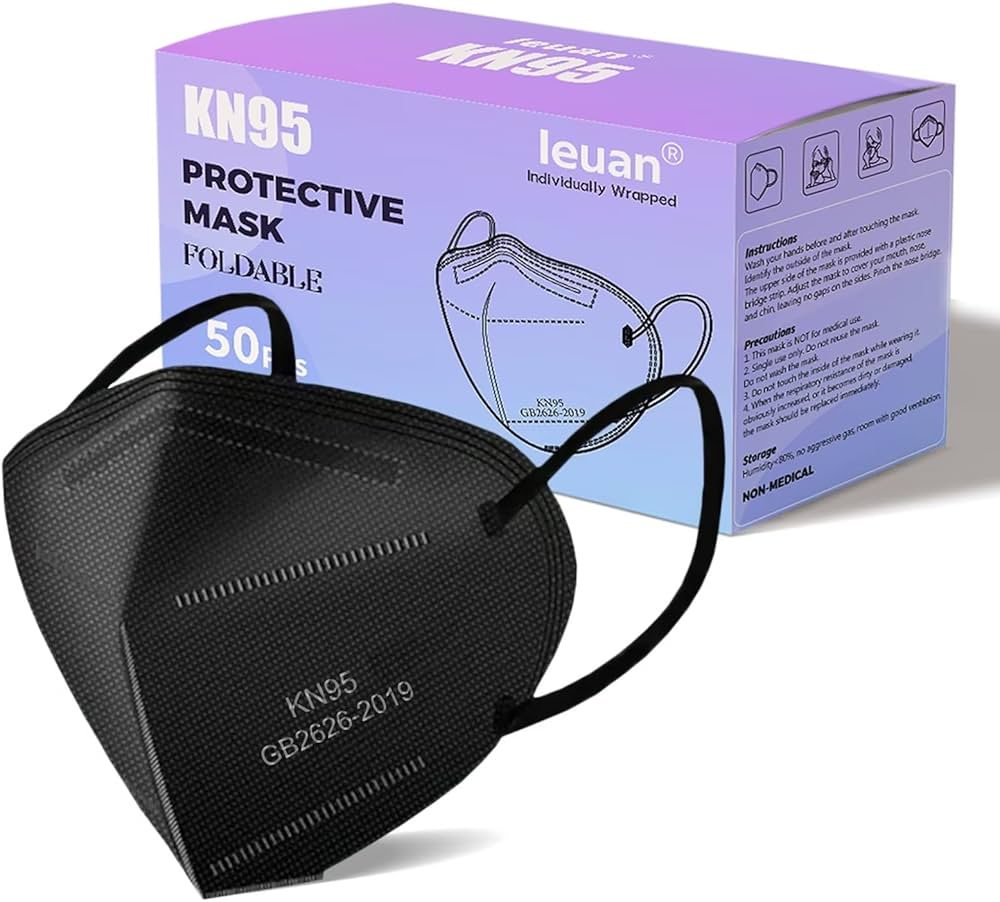
The KN95 Protective Respirator Mask is a high-filtration particulate respirator engineered to achieve at least 95% filtration efficiency against non-oil-based airborne particles, including dust, pollen, microbial droplets, and certain pathogenic aerosols. Its primary industrial use spans healthcare, pharmaceutical manufacturing, food processing, and heavy industrial settings where respiratory protection against particulates is mandated. The key value proposition is its dual compliance with rigorous international standards (GB2626-2019) and cost-optimized, scalable production—offering enterprises a reliable, auditable solution for workforce safety, regulatory compliance, and supply chain risk mitigation.
2. Key Specifications & Technical Characteristics
-
Material Composition: Multi-layer construction: 50–60 gsm spun-bond non-woven outer layer, high melt-blown polypropylene filter layer (≥99% bacterial filtration efficiency grade), hot-air cotton inner layer; nickel-free nose bridge; polyurethane or knit fabric ear loops.
-
Purity / Filtration Grade: KN95 grade per GB2626-2019; filtration efficiency ≥95% for 0.3 μm sodium chloride aerosol; differential pressure ≤ 85 Pa at 85 L/min airflow.
-
Physical Characteristics: Flat-fold, cup-less design; color: white (standard); weight: ~6–8 g per unit; dimensions (folded): ~155 × 105 mm (±5 mm); nose bridge length: ≥100 mm; adjustable.
-
Packaging Options: Individually sealed polybag (sterile or non-sterile); bulk inner pack: 10, 20, or 50 pcs per polybag; master carton: 1,000–2,000 pcs per carton (labels customizable with batch/lot number, expiration, and manufacturer ID).
-
Shelf Life: 3 years from date of manufacture when stored in original packaging at -20°C to +30°C, ≤80% relative humidity, away from direct sunlight and chemical fumes.
3. Core Industrial Applications
-
Primary Industries: Healthcare (clinics, labs, emergency services), pharmaceuticals (powder handling, compounding), food processing (facility sanitation, allergen containment), construction (masonry, insulation, demolition), mining, and general manufacturing (metal grinding, textile dust).
-
Specific Operational Use Cases:
-
Healthcare: Low-to-moderate risk patient contact, airborne precaution adjunct (when N95 not strictly mandated but KN95 approved under local regulation).
-
Pharmaceutical: Weighing and dispensing of non-hazardous powders; cleanroom grade B/D ancillary.
-
Food Processing: Mandatory PPE during deep cleaning cycles with airborne sanitizers or flour dust control.
-
-
Performance Advantage over Alternatives: Compared to surgical masks, KN95 achieves ~5x higher particulate capture; versus reusable elastomeric masks, it requires zero decontamination log, reducing operational labor by up to 90%. Durability tested to 40+ wear cycles (user fit-dependent). Cost per protected hour is 60–70% lower than certified N95 equivalents when sourced in bulk from compliant GB2626 vendors.
4. Competitive Advantages
-
Quality Consistency: ISO 13485:2016 certified manufacturing; each batch undergoes in-house filtration efficiency, inhalation/exhalation resistance, and flame retardancy testing per GB2626-2019, with third-party audit reports available upon request.
-
Supply Reliability: Vertically integrated production with 12 dedicated high-speed lines capable of 1.2 million units/day; buffer inventory of ≥30 days of raw melt-blown fabric to withstand upstream disruptions.
-
Logistics Capability: Global forward-stocking hubs (SE Asia, EU, US East Coast) enabling 7–10 day lead time to most major ports under standard terms.
-
Price Competitiveness: Direct factory pricing with no brand premium; volume tiers from 100k units downward adjust cost by ~15–20% versus distribution channels.
-
Sustainability & Documentation: Biodegradable outer layer options available; full traceability from resin lot to finished mask; user instruction sheets, declaration of conformity (DoC), and country-specific import permits provided as standard.
5. Commercial & Supply Information
-
Minimum Order Quantity (MOQ): BULK 20MT (metric tons) – equivalent to approximately 2.4–2.8 million individual masks depending on packaging density.
-
Loading Capacity: 20MT per standard 20-ft dry container (26–28 pallets), achieving full container load (FCL) optimization with zero wasted cubic volume. Mixed SKUs (different packaging or sterile/non-sterile) accepted within same container at proportion ≥10 MT per SKU.







Reviews
There are no reviews yet.