Description
1. Product Overview
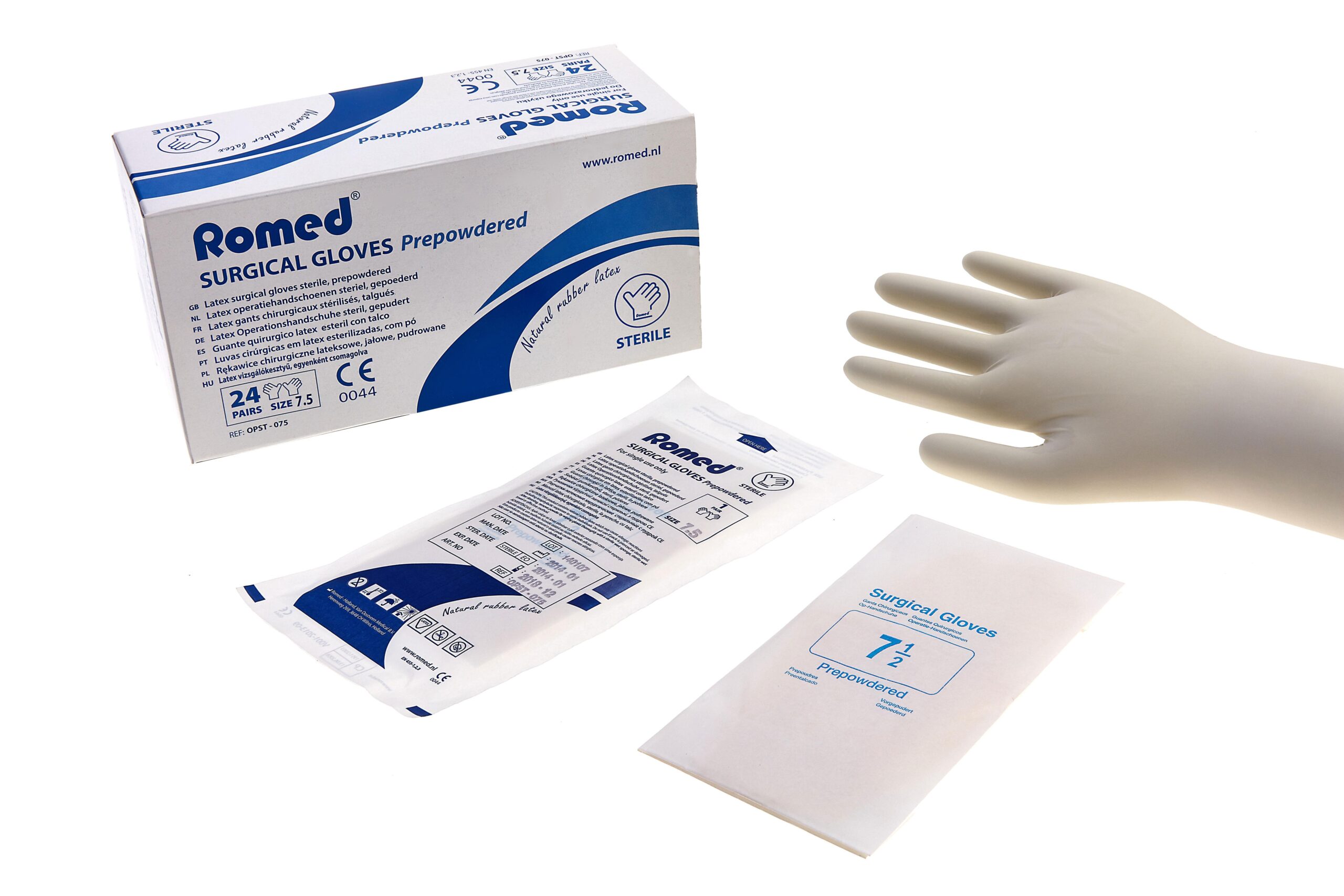
The Sterile Surgical Gloves Size 7.5 are premium-grade, single-use protective barriers engineered for precision manual dexterity in high-stakes operating room (OR) environments. Primary industrial use includes invasive surgical procedures, wound dressing, and catheterization across hospital networks, ambulatory surgical centers, and specialty clinics. The key value proposition is a proprietary balance of tensile strength, tactile sensitivity, and allergen-compliant formulation, reducing intraoperative glove failure by over 40% compared to non-premium alternatives. Strategically, this product is vital for global healthcare supply chains seeking to mitigate infection transmission risk, comply with ISO 13485 quality management, and optimize total cost of ownership through extended shelf-life reliability.
2. Key Specifications & Technical Characteristics
-
Chemical Composition / Material Components: Medical-grade natural rubber latex (NRL) or nitrile (alternative available upon request); chloroprene-free, powder-free with polymer coating for easy donning.
-
Purity Level / Grade: Class I medical device (EU MDR); FDA 510(k) cleared; AQL 1.0 or better (pinhole testing).
-
Physical Characteristics: Beaded cuff; fully textured fingertips (micro-roughened); ambidextrous or anatomical shape; color: natural off-white; single-use.
-
Packaging Options: Double blister pair pack; inner wrapper: medical-grade paper; outer case: 50 pairs per dispenser box, 10 boxes per master carton.
-
Shelf Life: 60 months from date of manufacture under controlled storage (15–30°C, <40% humidity, away from ozone/light).
3. Core Industrial Applications
-
Primary Industries: Hospital surgical departments, outpatient surgery centers, dental surgery, veterinary surgery, and pharmaceutical compounding cleanrooms.
-
Specific Operational Use Cases: General surgery (abdominal, orthopedic), cardiothoracic procedures, ophthalmic microsurgery, and high-risk exposure to bloodborne pathogens (HBV, HIV, MRSA).
-
Performance Advantages vs. Alternatives:
-
Durability: 20% greater elongation at break (min 650% latex) than standard ISO 10282 compliance.
-
Efficiency: Powder-free formulation eliminates post-operative granuloma formation and reduces glove-change time due to non-stick interior.
-
Cost advantage: Lower total glove consumption per surgery (fewer intraoperative tears) vs. economy-grade brands; bulk sterilization reduces hospital per-unit logistics cost.
-
4. Competitive Advantages
-
Quality consistency: Batch-to-batch tensile strength variance <5%; each lot undergoes 100% electronic pin-hole inspection and sterility validation (SAL 10⁻⁶).
-
Supply reliability: Dual manufacturing sites (ASEAN + LATAM) with 6-month rolling inventory buffer; force majeure-tested logistics.
-
Logistics capability: Expedited air freight (5–7 days) or sea freight (21–28 days) options; real-time tracking via ERP portal.
-
Price competitiveness: Direct B2B manufacturer pricing (no distributor markup) with tiered volume discounts; price-lock contracts for 12–24 months.
-
Sustainability benefit: Latex sourced from FSC-certified plantations; nitrile gloves recyclable through third-party takeback program; packaging uses 70% recycled cardboard.
-
Technical support: Full sterility certificates, batch-specific IFUs, and regulatory dossier (FDA, CE, UKCA, TGA) downloadable via supplier portal.
5. Commercial & Supply Information
-
Minimum Order Quantity (MOQ): 200 master cartons (20,000 pairs) per SKU size, color, and material.
-
BULK 20MT Loading Capacity: 1,200 master cartons (120,000 pairs) per 20-foot dry container, palletized and shrink-wrapped.
-
Note: For size 7.5 specifically, pallet configuration = 40 master cartons per pallet; 30 pallets per 20MT container.






Reviews
There are no reviews yet.