Description
Product Overview
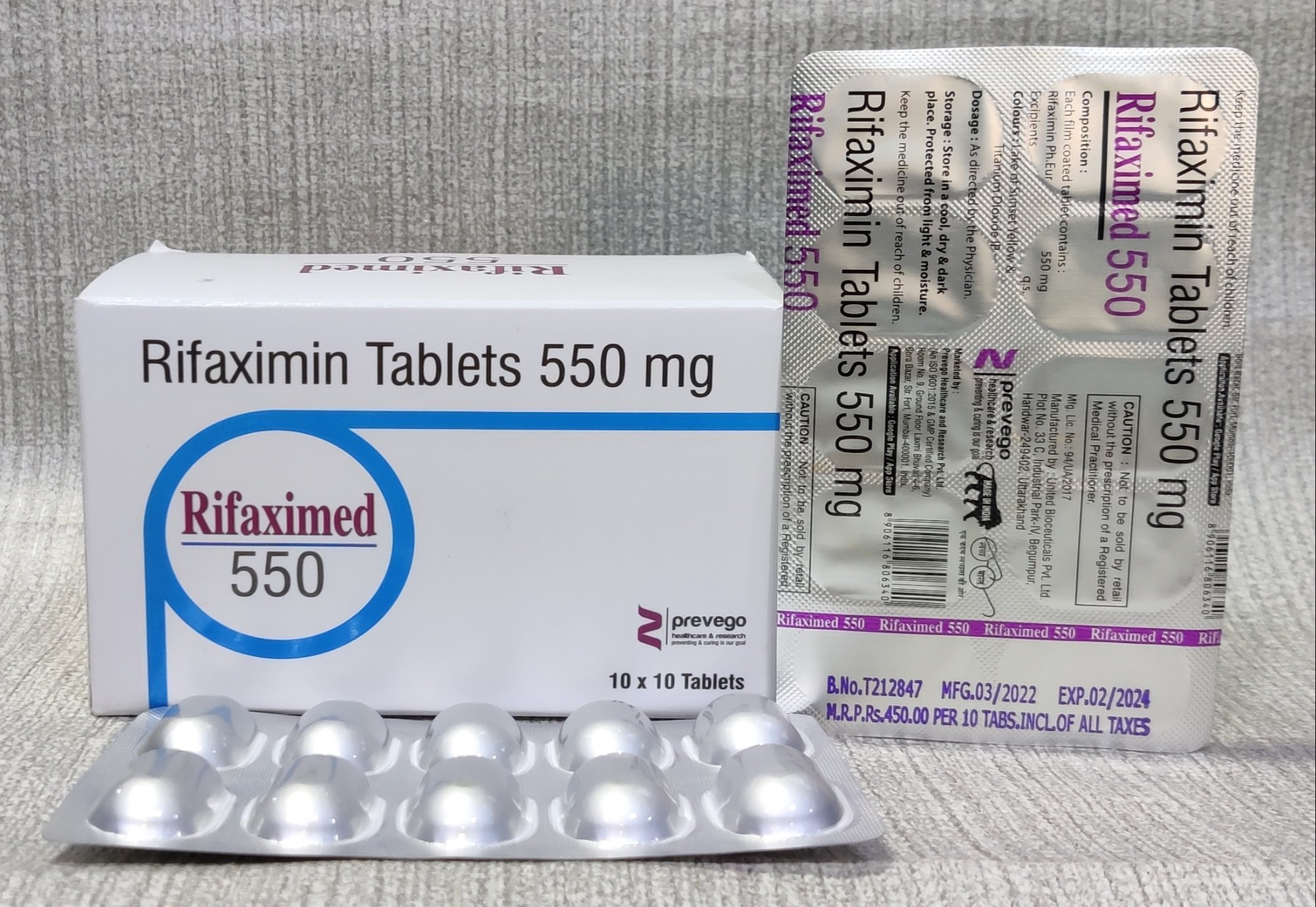
Rifaximin is a high-purity, semi-synthetic antibiotic derived from rifamycin, formulated for industrial and pharmaceutical-grade applications. It is primarily utilized for the production of oral formulations targeting gastrointestinal infections, including traveler’s diarrhea, hepatic encephalopathy prevention, and bacterial overgrowth disorders. Strategically, Rifaximin provides a superior efficacy profile with minimal systemic absorption, making it a preferred choice in global pharmaceutical manufacturing. Its stability, reproducible quality, and regulatory compliance ensure it is a critical asset for manufacturers aiming to maintain competitive advantage in the enteric antibiotic market. With strong industrial relevance, Rifaximin supports high-demand production lines and consistent product performance.
Key Specifications & Technical Characteristics
- Chemical Composition: C43H51N3O11
- Purity Level / Grade: ≥ 98% (Pharmaceutical/Industrial Grade)
- Physical Form: Fine crystalline powder
- Color: Off-white to pale yellow
- Particle Size: Micronized for uniform blending
- Density: Approx. 1.3 g/cm³
- Solubility: Practically insoluble in water; soluble in dimethyl sulfoxide (DMSO)
- Packaging Options: 25 kg kraft paper drums with inner polyethylene lining; custom packaging available upon request
- Shelf Life: 36 months under recommended storage conditions (cool, dry, and dark environment)
Core Industrial Applications
- Pharmaceutical Industry:
- Production of oral tablets, capsules, and powders for treating gastrointestinal infections.
- Used in formulations targeting traveler’s diarrhea, hepatic encephalopathy prevention, and irritable bowel conditions.
- Operational Advantages:
- Minimal systemic absorption ensures high local efficacy in the gut.
- Superior stability allows long shelf life and consistent dosage performance.
- Compatible with various excipients and scalable manufacturing processes.
- Efficiency & Cost Advantages:
- Reduces formulation variability and batch failure risk.
- High purity grade minimizes processing losses and downstream purification costs.
Competitive Advantages
- Quality Consistency: Stringent GMP-compliant production ensures uniform pharmaceutical-grade quality.
- Supply Reliability: Secure, scalable supply chain capable of meeting global industrial demand.
- Logistics Capability: Optimized for bulk shipment with compliant handling for pharmaceutical intermediates.
- Price Competitiveness: Competitive bulk pricing for high-volume industrial buyers.
- Sustainability & Compliance: Manufactured under environmentally responsible processes; supports green chemistry initiatives.
- Technical Support: Comprehensive documentation, including Certificates of Analysis (CoA) and regulatory compliance support.
Commercial & Supply Information
- Minimum Order Quantity (MOQ): BULK 20 MT
- Loading Capacity: 20 MT per 20-foot container (standard bulk shipping)
Rifaximin represents a strategic, high-value sourcing choice for industrial and pharmaceutical manufacturers seeking quality, reliability, and performance in enteric antibiotic production.







Reviews
There are no reviews yet.