Description
1. Product Overview
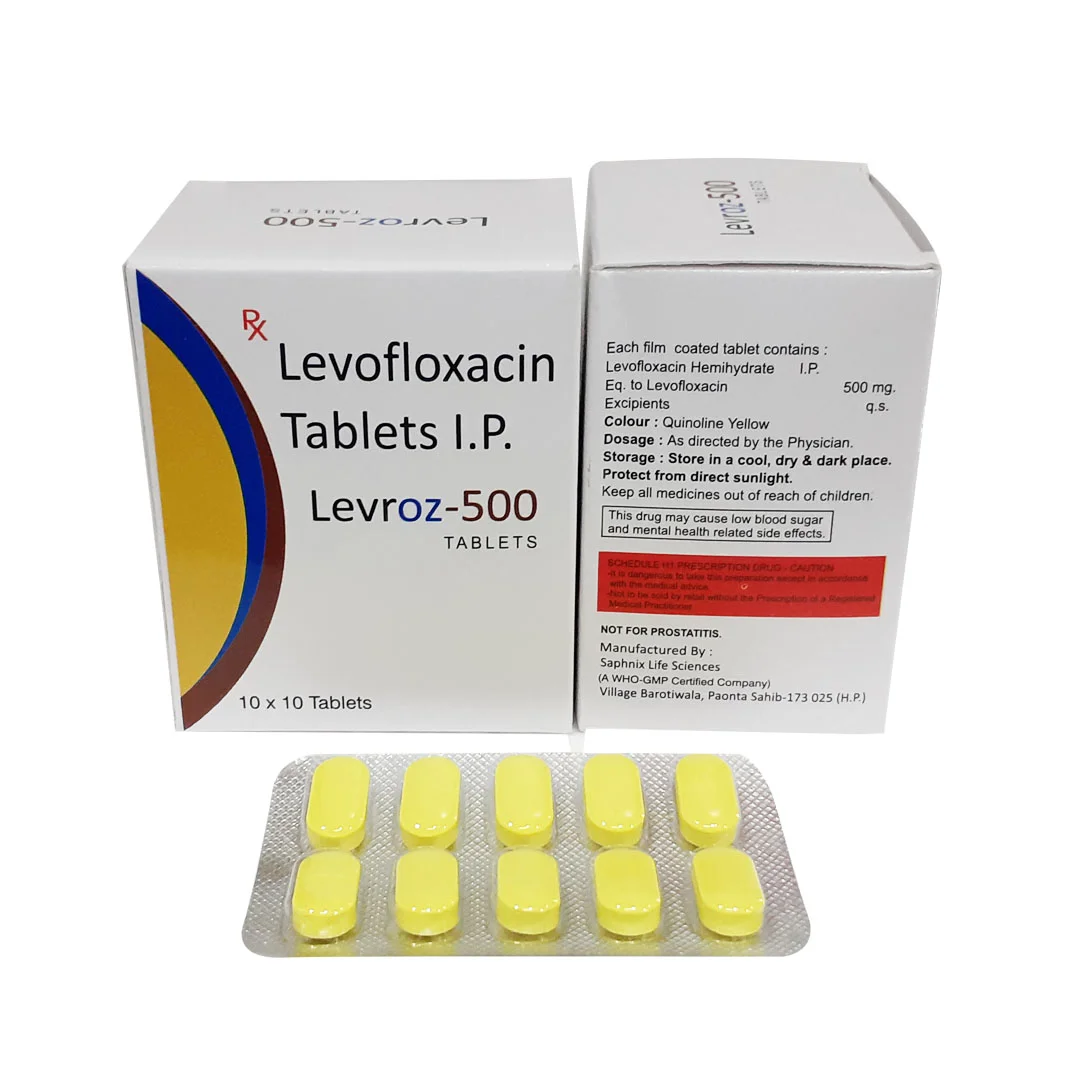
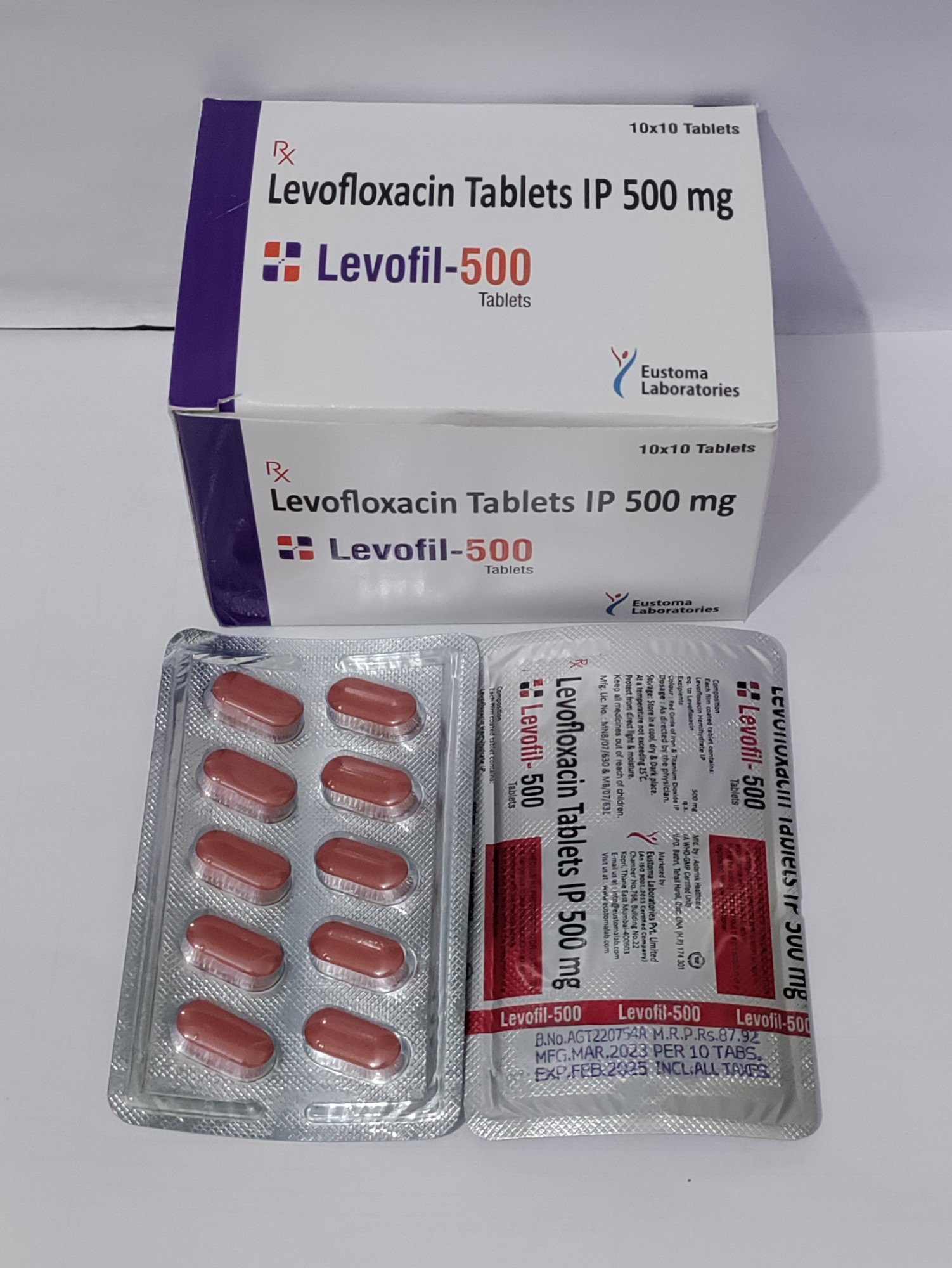
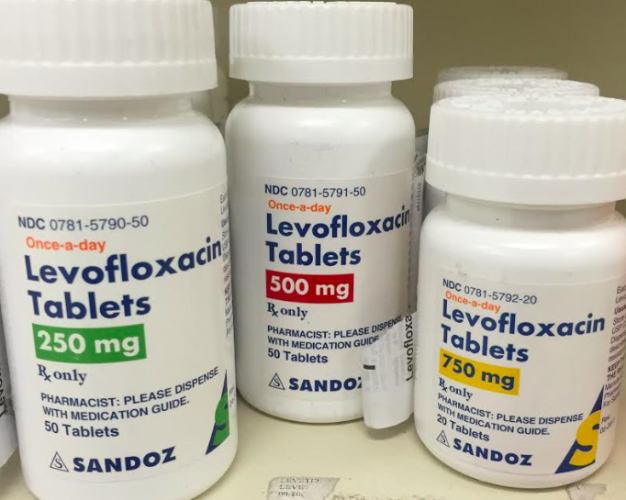
Levofloxacin is a high-potency, broad-spectrum fluoroquinolone antibiotic, widely recognized for its efficacy in inhibiting bacterial DNA gyrase and topoisomerase IV. Primarily utilized in pharmaceutical manufacturing, it serves as a critical active pharmaceutical ingredient (API) for oral and injectable formulations. Its superior bioavailability and stability make it a preferred choice for high-quality drug production, ensuring consistent therapeutic outcomes. Strategically, Levofloxacin holds a significant market position due to rising global demand for reliable antibacterial treatments and its inclusion in essential medicine portfolios. Industrial buyers value it for its predictable performance, regulatory compliance, and scalability in large-scale production.
2. Key Specifications & Technical Characteristics
- Chemical Composition: C₁₈H₂₀FN₃O₄ (Levofloxacin)
- Purity Level / Grade: ≥ 99% (Pharmaceutical Grade API)
- Physical Characteristics:
- Form: Crystalline powder
- Color: White to off-white
- Particle Size: 20–60 μm (adjustable on request)
- Bulk Density: ~0.45 g/cm³
- Solubility: Freely soluble in water at neutral pH; soluble in dilute acids
- Packaging Options: Multi-layered aluminum foil bags, drums, or ISO bulk containers
- Shelf Life: 36 months under recommended storage conditions (cool, dry, protected from light)
3. Core Industrial Applications
- Pharmaceutical Manufacturing: Used extensively in oral tablets, capsules, and intravenous formulations.
- Hospital & Clinical Supply Chains: Incorporated in antibiotic production for inpatient and outpatient care.
- Operational Advantages:
- High stability under varied manufacturing conditions
- Predictable dissolution rates ensuring consistent bioavailability
- Low impurity profile compared to alternative fluoroquinolones
- Performance Benefits: Enhances formulation efficiency, reduces batch-to-batch variability, and supports large-scale production without compromising quality.
4. Competitive Advantages
- Quality Consistency: Manufactured under stringent cGMP standards to meet global regulatory requirements.
- Supply Reliability: Robust production and logistics ensure uninterrupted supply for bulk orders.
- Technical Support: Comprehensive documentation, including COA, MSDS, and regulatory compliance certificates.
- Price Competitiveness: Optimized supply chain allows competitive pricing for large-volume procurement.
- Sustainability: Environmentally responsible production processes with minimized chemical waste and energy usage.
- Strategic Positioning: Ideal for pharmaceutical companies seeking dependable API sourcing for high-demand antibacterial drugs.
5. Commercial & Supply Information
- Minimum Order Quantity (MOQ): Bulk 20 MT
- Loading Capacity: 20 MT per 20-foot container
- Availability: Year-round production with scalable supply for industrial needs





Reviews
There are no reviews yet.