Description
1. Product Overview
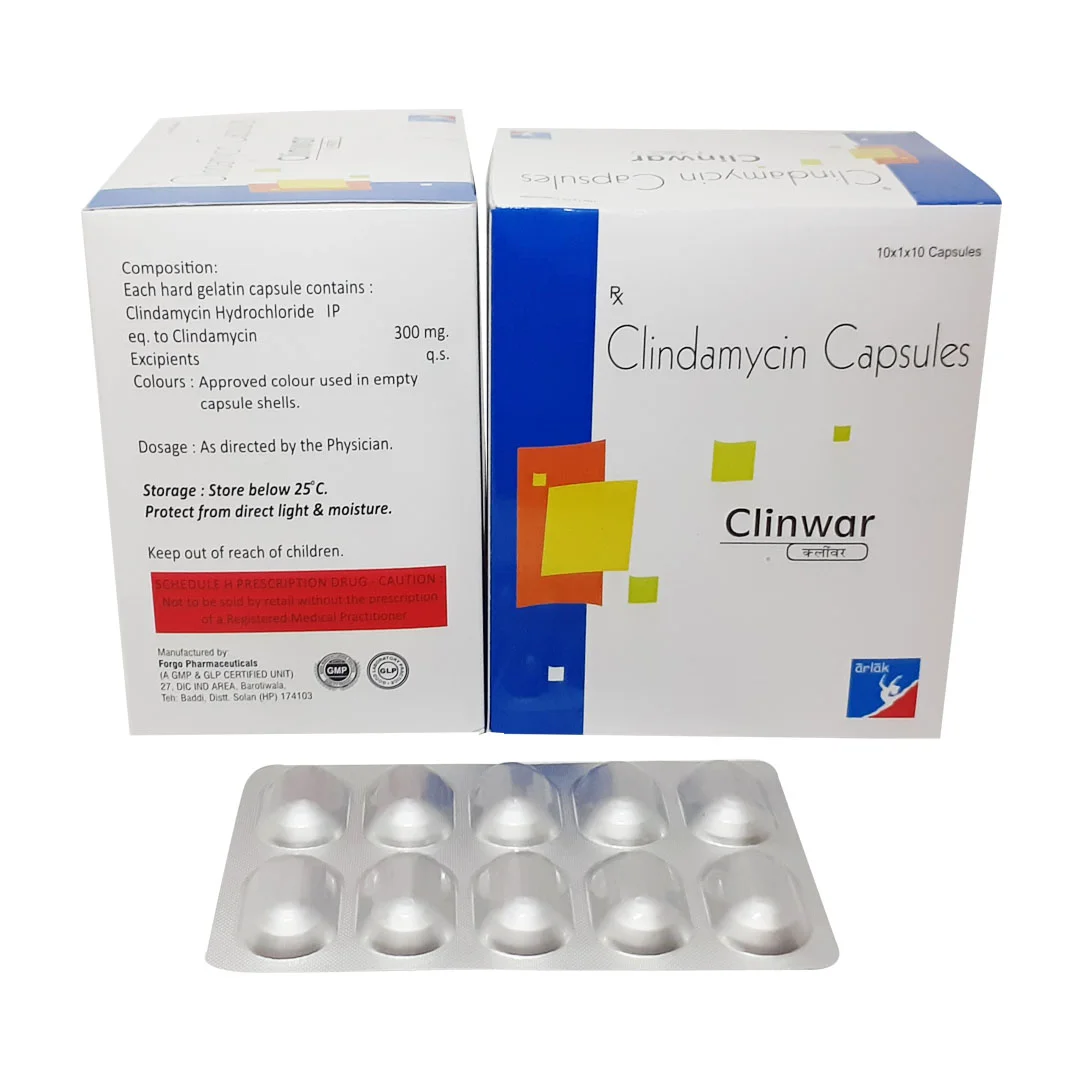
Clindamycin is a high-purity lincosamide antibiotic widely used in pharmaceutical manufacturing for systemic and topical formulations. It serves as a critical intermediate in producing oral capsules, injectables, and topical solutions, offering targeted antibacterial efficacy against a broad spectrum of Gram-positive pathogens. Strategically, Clindamycin ensures consistent supply for essential drug production, making it a key ingredient in global healthcare supply chains. Its reliability, regulatory compliance, and proven pharmacological profile position it as a high-value sourcing choice for pharmaceutical manufacturers worldwide.
2. Key Specifications & Technical Characteristics
- Chemical Composition: Clindamycin hydrochloride (or base, per buyer specification)
- Purity / Grade: ≥98% pharmaceutical grade
- Physical Form: White to off-white crystalline powder
- Solubility: Soluble in water, methanol, and ethanol
- Particle Size: Fine powder suitable for uniform dissolution
- Density: ~1.3 g/cm³ (bulk density)
- Moisture Content: ≤1.0%
- Packaging Options: 25 kg fiber drums with double-layered inner polyethylene liners or customized per client requirements
- Shelf Life: 24 months under recommended storage conditions (cool, dry, away from light)
3. Core Industrial Applications
- Primary Industries:
- Pharmaceutical manufacturing
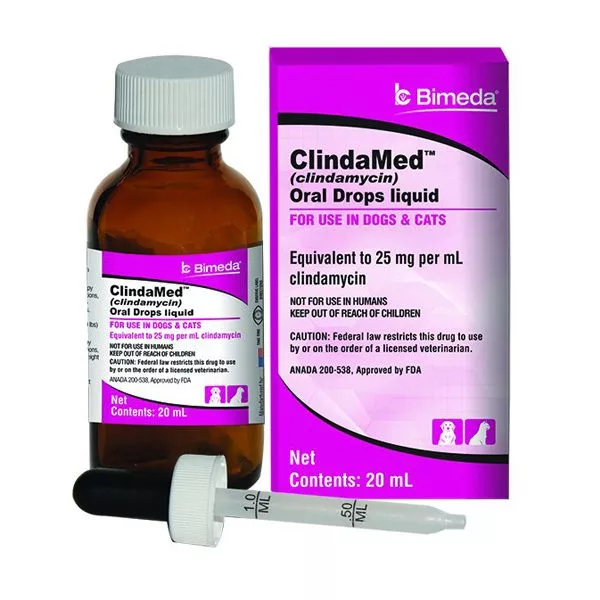
- Veterinary medicine production
- Operational Use Cases:
- Production of oral capsules, tablets, and injectables
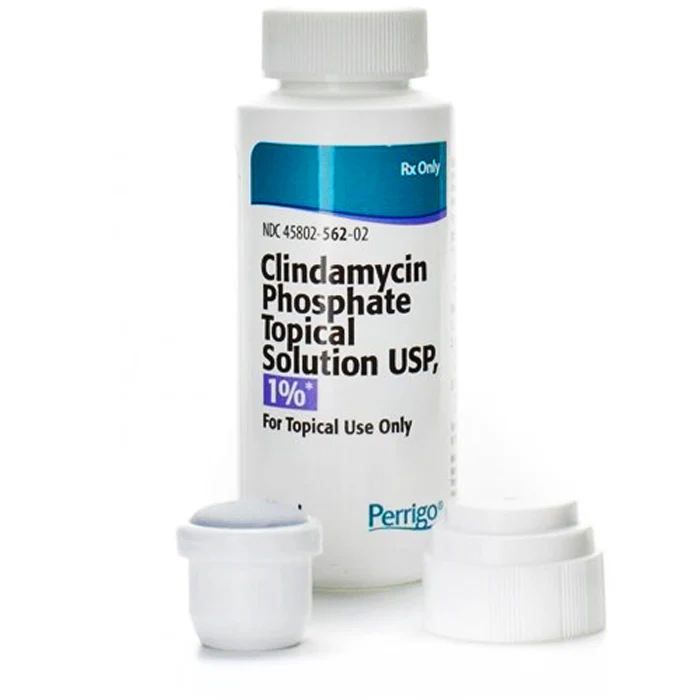
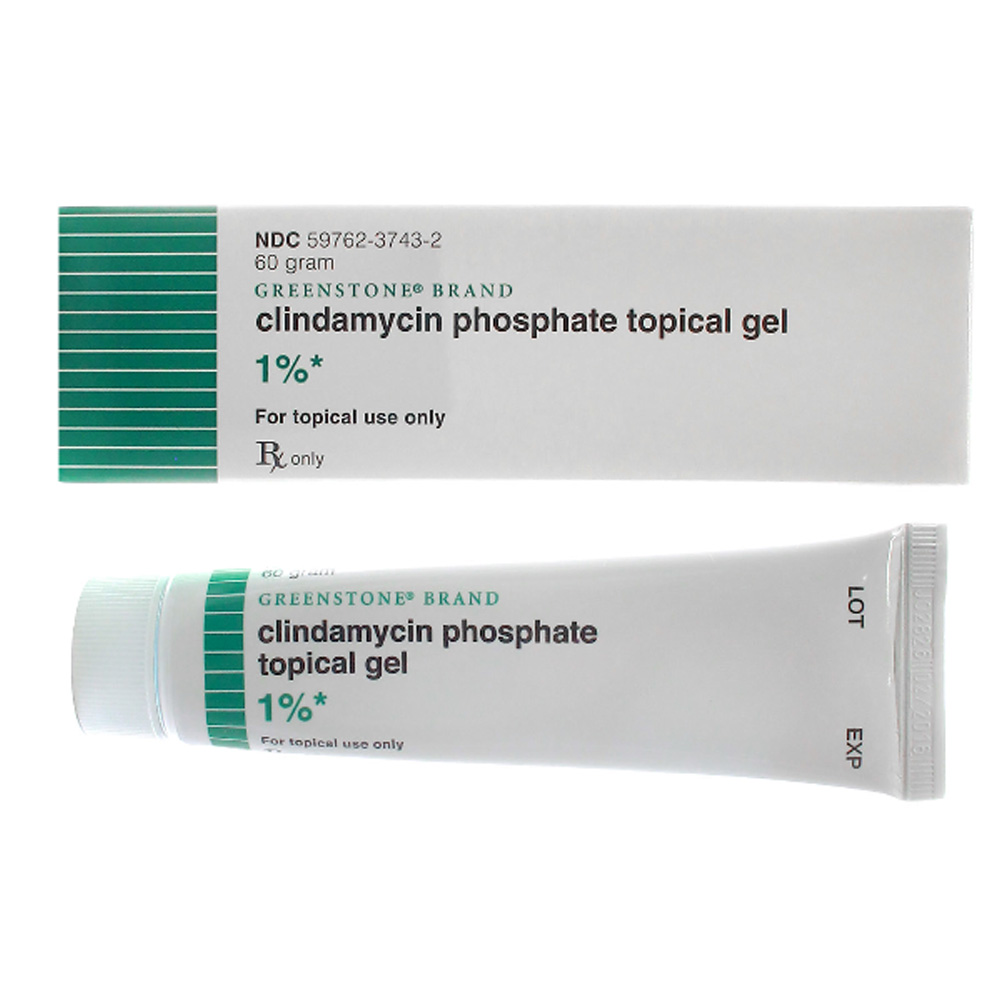
- Formulation of topical gels, creams, and solutions
- Performance Advantages:
- High stability under standard manufacturing conditions
- Superior solubility for efficient formulation
- Consistent batch-to-batch quality reduces process variability
- Commercial Relevance:
- Enhances production efficiency through reliable dissolution and bioavailability
- Supports compliance with global regulatory standards (USP, EP, JP)
4. Competitive Advantages
- Quality Consistency: Strict GMP-compliant manufacturing ensures uniformity across batches
- Supply Reliability: Robust production capacity secures uninterrupted supply
- Logistics Capability: Flexible shipping options, including containerized bulk and specialized packaging
- Price Competitiveness: Optimized production allows cost-effective bulk sourcing
- Sustainability: Environmentally responsible production methods and waste management practices
- Technical Support: Comprehensive documentation, including Certificates of Analysis (CoA) and regulatory compliance data
5. Commercial & Supply Information
- Minimum Order Quantity (MOQ): 20 MT (BULK)
- Loading Capacity: 20 MT per standard 20-foot container











Reviews
There are no reviews yet.