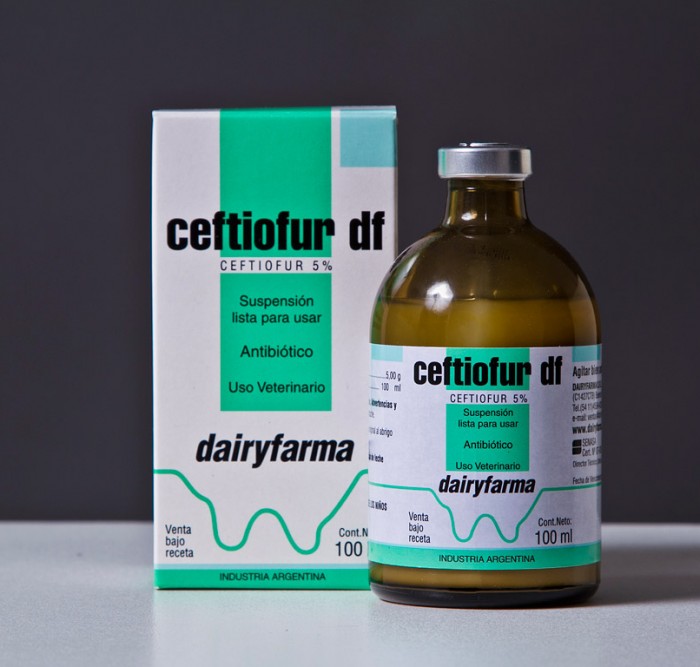
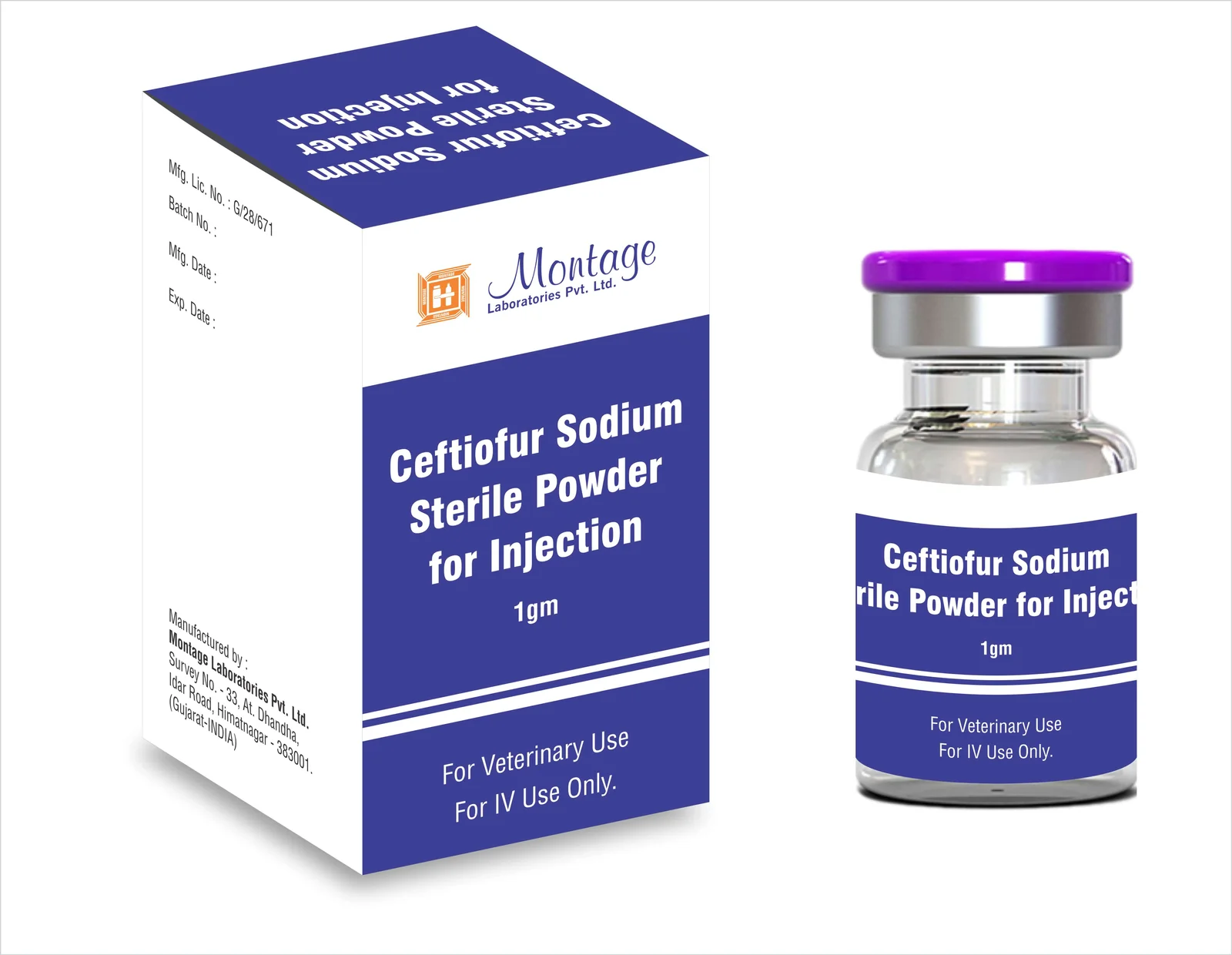
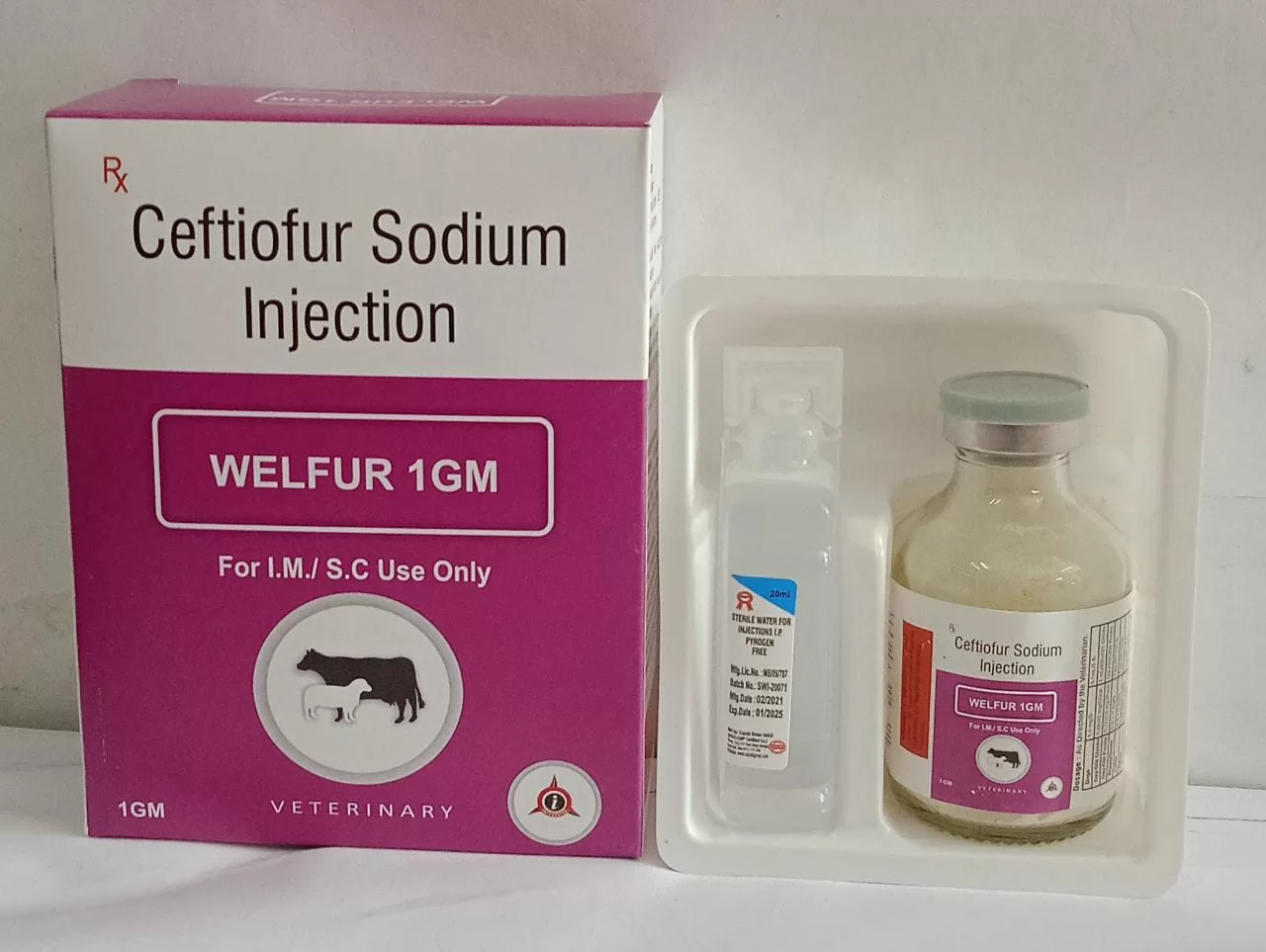
Description
1. Product Overview
Ceftiofur is a broad-spectrum cephalosporin antibiotic widely used in veterinary medicine for the prevention and treatment of bacterial infections in livestock. Its primary industrial use is in the formulation of injectable and oral veterinary products, targeting respiratory, enteric, and systemic bacterial diseases in cattle, swine, and poultry. The key value proposition of Ceftiofur lies in its high efficacy, rapid action, and favorable pharmacokinetic profile, ensuring improved herd health and productivity. Strategically, it is important in the market as a critical veterinary antimicrobial that supports disease control, reduces economic losses from livestock infections, and aligns with regulatory standards for animal health. Its professional-grade formulation ensures consistent performance and reliability for large-scale veterinary operations.
2. Key Specifications & Technical Characteristics
- Chemical Composition / Material Components:
- Active pharmaceutical ingredient: Ceftiofur (C17H17N5O7S3)
- Excipients may vary depending on formulation
- Purity Level / Grade:
- Pharmaceutical grade, ≥ 98% purity
- Conforms to veterinary pharmacopoeia standards (e.g., USP, EP)
- Physical Characteristics:
- Form: Powder or crystalline solid
- Color: White to off-white
- Particle size: Fine crystalline
- Density: Batch-specific; typically 0.6–0.8 g/cm³
- Packaging Options:
- Bulk drums
- Polyethylene-lined steel containers
- Custom veterinary packaging upon request
- Shelf Life:
- Typically 24–36 months under recommended storage conditions
- Store in a cool, dry place away from light and moisture
3. Core Industrial Applications
Primary Industries:
- Veterinary pharmaceutical manufacturers
- Livestock health management operations
- Feed additive and animal health integrators
Specific Operational Use Cases:
- Formulation of injectable solutions for cattle, swine, and poultry
- Integration into oral powders or premixes for preventive care
- Used in veterinary clinics and large-scale livestock farms for bacterial infection control
Performance Advantages:
Ceftiofur outperforms many alternatives due to its broad-spectrum activity, rapid absorption, and long-lasting therapeutic effect. Its stability and predictable pharmacokinetics enhance treatment efficacy, minimize dosing frequency, and improve operational efficiency in livestock management.
4. Competitive Advantages
- Quality Consistency: Strict batch-level quality control and analytical testing
- Supply Reliability: Continuous production with established veterinary API sources
- Logistics Capability: Compliant transportation of pharmaceutical-grade material
- Price Competitiveness: Cost-effective alternative to imported APIs
- Sustainability Benefits: Supports responsible veterinary practices and herd health management
- Technical Support: COA, SDS, pharmacopoeia compliance, and regulatory documentation available
Ceftiofur is positioned as a strategic sourcing decision for veterinary pharmaceutical companies seeking reliable, high-quality, and regulatory-compliant antibiotic APIs.
5. Commercial & Supply Information
- Minimum Order Quantity (MOQ): BULK 20MT
- Loading Capacity (MT per container): Approximately 20 MT per 20’ ISO tank container (subject to packaging and density considerations)













Reviews
There are no reviews yet.